Introduction
Cardiovascular complications develop frequently in patients with coronavirus disease 2019 (COVID-19) [1]. Acute coronary syndromes (ACS) remained the leading cause of death and disability all over the world [2, 3]. Systemic viral infections may generate plaque rupture and thrombosis [4, 5]. Notably, patients with COVID-19 and ACS have rarely been described in reports [6–9].
All ST-segment elevation myocardial infarction (STEMI) patients should undergo an early assessment of both short-term risk on admission and long-term risk at discharge [10]. Selecting the group of patients with an unfavorable prognosis makes it possible to take actions that can potentially prevent the occurrence of adverse events. These actions include prolonged monitoring of basic vital parameters such as electrocardiogram (ECG), SaO2, blood pressure or delayed discharge after myocardial infarction. Prognostic scoring systems including the Global Registry of Acute Coronary Events (GRACE) risk score and thrombolysis in myocardial infarction (TIMI) risk score have been reported to be useful in predicting in-hospital and long-term mortality among patients with ACS [11, 12]. There are eight parameters used to calculate the Global Registry of Acute Coronary Events (GRACE) risk score – age, heart rate, systolic blood pressure, initial creatinine level, Killip class, pre-hospital cardiac arrest, elevated cardiac markers and ST-segment elevation or depression – which are evaluated to calculate the risk of in-hospital mortality [11, 13]. The original GRACE data did not include patients with concomitant SARS-CoV-2 infection.
Sparse reports of cardiac catheterization in patients with concomitant COVID-19 reveal a variety of angiographic findings in patients with ACS including obstructive coronary disease (type 1 myocardial infarction), angiographically normal epicardial arteries (e.g. stress-induced cardiomyopathy) or high thrombus burden [14, 15]. In others, fibrinolytic therapy was the main treatment strategy so there is no information regarding coronary arteries [16].
As there are limited reports on patients with STEMI and COVID-19 treated with percutaneous coronary intervention (PCI), we aimed to describe their demographic, angiographic and periprocedural characteristics along with their clinical outcomes. Moreover, the present study aimed to examine the association between the GRACE risk score and in-hospital mortality in this group of patients, which, to our best knowledge, has not been done so far.
Material and methods
This was a single-center, retrospective study of 25 patients admitted to a multi-specialist hospital where a cardiology ward for COVID-19 patients was established. The ward existed from October 23, 2020 to April 23, 2021 (exactly 6 months). Anonymized data were gathered retrospectively. This study was designed to assess the mortality rate in the group of patients with both STEMI and COVID-19 and to compare baseline characteristics, the angiographic view, and procedural and clinical outcomes between survivors and non-survivors. The analyzed endpoint was in-hospital mortality from any cause. None of the patients had been fully vaccinated against severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2). Patients were diagnosed as having COVID-19 infection through positive results on PCR testing of nasopharyngeal samples in the post-catheterization period. STEMI was defined based on the presence of typical chest pain combined with ST-segment elevation in a 12-lead electrocardiogram following the fourth universal definition of myocardial infarction [17].
Both comorbidities and risk factors were identified based on medical history or prior diagnosis or treatment and defined based on the up-to-date ESC guidelines [18].
Laboratory parameters
Blood samples were obtained on admission before cardiac catheterization. Blood cell count, glucose, creatinine, high-sensitivity troponin I (hs-TnI), procalcitonin (PCT) and lipid profile were measured using routine laboratory techniques. The blood count was measured by a Siemens high volume hematology analyzer ADVIA 2120i. A sodium citrate tube and ACL TOP 500 analyzer were used for quantitative D-dimer measurement. High-sensitivity C-reactive protein (hsCRP) was measured quantitatively by immunoturbidimetric assay (Abbott Alinity C, Illinois, U.S.A.).
Interventional procedures
During the study period, in the case of confirmed or suspected COVID-19 positive patients medical personnel wore full personal protective equipment during contact.
Obstructive coronary artery disease was defined as the presence of significant narrowing of the vessel > 50%. All patients underwent immediate coronary angiography and no one was treated with fibrinolysis.
The interventional strategy, including the use of direct stenting, balloon pre- and post-dilatation, aspiration thrombectomy, the use of glycoprotein (GP) IIb/IIIa inhibitors and the type of stent, was at the discretion of the operator and according to guidelines. All subjects were administered a loading dose of aspirin 300 mg and either clopidogrel (600 mg), ticagrelor (180 mg) or prasugrel (60 mg). Maintenance therapy consisted of a daily dose of 75 mg of aspirin and either clopidogrel (75 mg), ticagrelor (90 mg twice a day) or prasugrel (10 mg). During primary PCI unfractionated heparin was given intravenously in a loading dose of 70 to 100 U/kg or 50 to 70 U/kg in the case of glycoprotein IIb/IIIa (GP IIb/IIIa) inhibitor use.
Assessment of left ventricular ejection fraction (LVEF) was performed using the echocardiographic biplane Simpson method.
Prediction of in-hospital mortality was based on the GRACE risk score and the calculation was performed by identifying and summing the scores for each predictive factor [11].
Statistical analysis
Data were analyzed using PQStat v.1.8.2.232 Software (Poznan, Poland). Categorical variables were expressed as numbers and percentages. The Student t-test (the number of degrees of freedom (df) was 23) was used for variables with normal distribution (Shapiro-Wilk test) and the values were presented as mean ± SD. Others were analyzed using the Mann-Whitney U test and expressed as median with IQR (25th and 75th). Qualitative data were compared by the χ2 (the number of df was 1) and Fisher’s exact test. The association between parameters was assessed by Spearman’s rank correlation test. Receiver operating characteristics (ROC) curve and area under the curve (AUC) with optimal cut-off were used to analyze the discriminatory power of the GRACE scale. A two-tailed p-value < 0.05 indicates statistical significance and the confidence interval (CI) was 95%.
Results
Patient characteristics
The study population consisted of 25 consecutive patients with confirmed STEMI who were admitted during 6 months. From October 23rd 2020 to April 23rd 2021 they were admitted to the cardiological ward and completed their hospital stay (i.e. discharge or death). In each case, a positive result was obtained on the first day of stay; hence they were defined as community-acquired infections.
The demographic and clinical characteristics of the studied groups are presented in Table I.
Table I
Characteristics of the studied groups
[i] Data are given as mean ± standard deviation, median (interquartile range) or number (percentage). BMI – body mass index, ECG – electro-cardiogram, GRACE – Global Registry of Acute Coronary Events, hsCRP – high-sensitivity C-reactive protein, hs-TnI – high-sensitivity troponin I, LDL-C – low-density lipoprotein cholesterol, LVEF – left ventricular ejection fraction, NT-pro-BNP – N-terminal prohormone of brain natriuretic peptide.
The median age was 70 years and 92% were male. 100% of them were of the Caucasian race. There were no significant differences in age, sex or body mass index (BMI) between the two groups. The median hospitalization duration was longer in the survivor group than in non-survivors (median (Q1–Q3); 17 (13–21) vs. 8.5 (3.5–14.5) days, respectively, p = 0.026). Total mortality was 48% (12 of 25 subjects, of whom 75% were older than 65 years (9 of 12)).
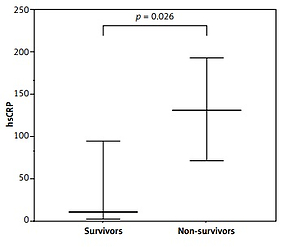
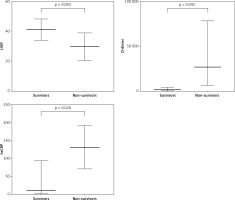
Despite higher rates of mechanical ventilation in the non-survivor group, this did not reach significance (67% vs. 46%, p = 0.3). It is worth noting that there was a tendency (with borderline significance) to higher prevalence of cardiogenic shock in the non-survivor group (50% vs. 15%, p = 0.097). The number of pre-hospital cardiac arrests was comparable in both groups. Reduced LVEF was observed in the non-survivor group (mean ± SD; 30 ± 9 vs. 41 ± 7, p = 0.003) (Figure 1).
Figure 1
Comparison of selected parameters. Medians and interquartile ranges (D-dimer and hs-CRP) or means and standard deviations (only LVEF) are presented
No difference between the groups was observed concerning the ECG presentation. The most common was an infarction of an anterior wall (52%).
The most widespread cardiovascular risk factor or disease was arterial hypertension (68%), followed by diabetes mellitus (48%) and obesity (40%).
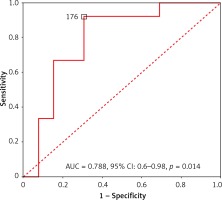
There was a higher GRACE score in the non-survivor group (mean ± SD; 210 ±35 vs. 169 ±42, p = 0.014). In ROC analysis, GRACE score predicted in-hospital death with an AUC of 0.788 (95% CI: 0.6–0.98, p = 0.014) (Figure 2). A score of 176 was identified as the optimal cut-off with a sensitivity of 92% and specificity of 69%. Of note, only one patient with in-hospital death had a GRACE score < 176 in the present study.
Figure 2
ROC curve of the total GRACE risk score for in-hospital mortality. Optimal cutoff and AUC (with 95% CI) are presented
Laboratory parameters on admission are presented in Table I. Non-survivors had a higher level of hsCRP (median (Q1–Q3); 131 (72–190) vs. 13 (4–96), p = 0.026) and D-dimer (median (Q1–Q3); 27327 (5070–78476) vs. 652 (474–1498), p = 0.042) (Figure 1).
GRACE score was positively correlated with procalcitonin (r = 0.604, p = 0.004), whereas it was negatively correlated with LVEF (r = –0.498, p = 0.016). D-dimer was positively correlated with N-terminal prohormone of brain natriuretic peptide (NT-pro-BNP) (r = 0.794, p = 0.0061).
Procedural characteristics and treatment
A primary PCI procedure was performed in all patients in both groups (Table II). Median door-to-balloon times were well below 60 min and comparable in both groups. Access through the radial artery was the most commonly used one. We found no differences with regard to the culprit artery. The left anterior descending artery was the most often responsible for myocardial infarction (44%) followed by the right coronary artery (36%) and circumflex artery (16%). Similar levels of baseline TIMI flow grade, the rate of aspiration thrombectomy use and successful stent implantation were observed in both groups. Postprocedural TIMI 3 was slightly less frequently observed in the non-survivor group (p = 0.039); however, this did not directly translate into more frequent use of GP IIb/IIIa inhibitors (p = 0.64). Only second-generation stents were implanted, of which the most common antiproliferative substance in both groups was sirolimus (48%).
Table II
Procedural characteristics and treatment
[i] Data are given as median (interquartile range) or number (percentage). 3VD – three-vessel disease, ACEI – angiotensin-converting enzyme inhibitor, ARB – angiotensin II receptor blocker, ASA – acetylsalicylic acid, Cx – circumflex artery, DES – drug-eluting stent, GP IIb/IIIa – glycoprotein IIb/IIIa, LAD – left anterior descending artery, LMS – left main stem, RCA – right coronary artery, TIMI – thrombolysis in myocardial infarction.
Pharmacotherapy is presented in Table II.
The most commonly used P2Y12 inhibitor in combination with aspirin was clopidogrel (68%) followed by ticagrelor (28%). Β-blockers and angiotensin-converting enzyme inhibitors (ACEIs)/angiotensin II receptor blocker (ARBs) were used less often by non-survivors (p = 0.002 and p = 0.015; respectively), whereas the use of catecholamines was the opposite (p = 0.028).
Discussion
The current study was the first to assess the GRACE risk scoring system suitability for individuals with SARS-CoV-2 infection. We found a strong correlation between the GRACE risk score and short-term death despite the small sample. A score ≥ 176 indicated a high risk for patients with STEMI. Another factor associated with mortality in this group is decreased LVEF as assessed by transthoracic echocardiography, which is in line with other observations (in non-COVID-19 patients); however, it was not taken into account when projecting the GRACE risk score [19, 20]. The overall mortality in this study was 48%, which is lower than in a smaller case series in New York (72%) and slightly higher than was reported in a case series in Italy (39.3%, 3.6% still hospitalized) [8, 9]. When comparing these groups it is worth noting that our patients were older and had a greater number of comorbidities.
COVID-19 infection is associated with a prothrombotic state [21]. The presence of venous thromboembolism is related to disease severity and clinical outcome among patients with COVID-19 [22]. We found that non-survivors had higher levels of high-sensitivity C-reactive protein and D-dimers which reflect a systemic inflammatory response and prothrombotic state. These parameters were also found to be independently associated with a higher mortality rate in non-COVID-19 patients with STEMI [23, 24]. Choudry et al. reported that there is strong evidence for a higher load of thrombus in subjects presenting with STEMI and concomitant COVID-19 infection reflected by higher thrombus grade, more frequent use of aspiration thrombectomy and GP IIb/IIIa inhibitor when compared with non-COVID-19 patients [15]. When comparing groups of patients with myocardial infarction and SARS-CoV-2 in both our and the above-mentioned study, similar frequency was observed with regard to the use of aspiration thrombectomy (20% in our study vs. 17.9%); however, the use of GP IIb/IIIa in our group was less frequent (20% vs. 59%) but still higher than in non-COVID-19 patients (9.2%) [15]. Surprisingly, the most frequently chosen P2Y12 inhibitor in our study was clopidogrel (68%), which cannot be explained by the need for anticoagulation due to the risk of thromboembolism in the course of atrial flutter or fibrillation (28%). However, due to numerous reports of effectiveness in the prevention of major thromboembolic events and death, therapeutic doses of heparin were widely used in our group [25]. In this case, the use of prasugrel or ticagrelor could have led to major bleeding compared to clopidogrel [26, 27]. In some cases, clopidogrel was chosen by the patient due to financial reasons dictated by the large discrepancy in the purchase prices.
Unlike other reports, all of our patients were treated with primary percutaneous coronary intervention [16]. None of them was treated with fibrinolytic therapy. The optimal management strategy for patients with STEMI and coexisting COVID-19 was a substantial challenge at the beginning of the pandemic. In order to mitigate the spread of the disease, before the mechanisms of transmission of the infection were fully understood, fibrinolysis was a procedure recommended by Chinese experts in stable individuals who presented within 12 h of symptom onset and did not have any contraindications [28]. However, primary PCI remained generally the standard of care for STEMI patients when it could be provided in a timely fashion [29].
As regards pharmacotherapy, Β-blockers and ACEIs/ARBs were used less often by non-survivors. There is a confirmed correlation between chronic ACEI exposure and a milder course of COVID-19 [30]. However, it is worth noting that there was higher prevalence (though not significant) of cardiogenic shock in the non-survivor group, and this contraindication in drug use could translate to the differences we observed.
It is a relatively small, retrospective, monoethnic, observational study in a single center and therefore has all the limitations of this kind of analysis. Although there is no intergroup difference with regard to sex, women are underrepresented. Female sex is an independent predictor of hospital mortality in the STEMI population; however, it does not substantially improve the discriminative capacity of the GRACE risk score [31]. Despite the limitations, as far as we know, this is the first study aiming to assess the GRACE risk score in individuals with COVID-19 and STEMI. The clinical relevance of our findings needs to be further explored in a larger number of patients in a multicenter study.
In conclusion, as far as we know, this is the first study to demonstrate a significant correlation between the GRACE risk score and short-term mortality in the population with STEMI and concurrent COVID-19. Among other factors, non-survivors had elevated hsCRP and D-dimer and reduced LVEF. Postprocedural TIMI 3 flow grade was less frequently observed in this group. Nonetheless, our results need to be validated by larger studies in the future.