Introduction
Obstructive sleep apnea (OSA) is strongly linked with several forms of cardiovascular diseases with a high prevalence in the general population and even higher prevalence among patients with known cardiovascular diseases [1]. Several disturbances caused by OSA may provoke various systemic imbalances such as hypoxemia which can trigger various complications, including conduction abnormalities [2]. The diagnosis and timely treatment of OSA is also believed to be of crucial importance in preventing acute myocardial infarctions (AMI) and acute ischemic strokes (AIS) [3, 4]. However, as little is known about the patient characteristics and different cardiac arrhythmias seen in OSA patients following an AMI or AIS, the 2019 National Inpatient Sample (NIS) was used to analyze and study the incidence and outcomes of these patients [5].
Material and methods
The NIS is a yearly set of databases developed and maintained by the Healthcare Cost and Utilization Project (HCUP), the Agency for Healthcare Research and Quality (AHRQ), and their many collaborators. It contains more than 7 million hospitalization records in the “unweighted” form, and in its “weighted form” it can cover > 35 million patients over a year. The 2019 NIS is the most recent version released by the group, and several changes have been made to improve the accuracy of the databases over the last few years. Additional information can be obtained at https://www.hcup-us.ahrq.gov/nisoverview.jsp [5].
Patients aged 25 and over were retained for the study. Our sample was also converted to its “weighted” form, which, as per the HCUP guidelines, allowed us to project a national estimate of various conditions. Principal diagnoses of AMI and AIS were obtained, and patients with a diagnosis of OSA at any diagnostic level were also identified. Our study was divided into two steps. First, we estimated the patient characteristics to understand the differences between OSA and non-OSA groups for patients having been hospitalized for AMI or AIS. We also identified various cardiac arrhythmias (supraventricular tachycardia, ventricular tachycardia, paroxysmal atrial fibrillation, and ventricular fibrillation). We then used multiple regression analysis to estimate the adjusted odds ratio (aOR) of an OSA patient dying during their hospitalization and the impact of various possible risk factors on their mortality risks [5].
HCUP data consist of de-identified and anonymous patients, and the needs of ethic clearance and IRB approval are waived as per their data use agreement and guidelines [5].
Results
551,385 patients had a principal diagnosis of AIS, with 39,370 (7.1%) also having a diagnosis of OSA (Table I). A higher incidence was reported among males than females (8.8% of males with AIS also had OSA, while 5.4% of females with AIS also had OSA, p < 0.01). While most AIS patients were aged 66 or over (58.6%), patients aged 46–65 with AIS were also more likely to have a diagnosis of OSA than the other two age groups (8.4% among 46–65-year-olds, p < 0.01). OSA was also more common among Whites admitted for AIS (7.8% of Whites with AIS had OSA, p < 0.01). 42.9% of OSA patients with AIS were obese (p < 0.01), while 91.2% had hypertension (p < 0.01), and 53.5% had diabetes (p < 0.01). A higher incidence of ventricular tachycardia (2.1%) and paroxysmal atrial fibrillation was recorded among patients with OSA (p < 0.01 each), while OSA patients reported a slightly lower incidence of supraventricular tachycardia (1.3%) than non-OSA groups (1.4%).
Table I
Characteristics of obstructive sleep apnea patients following an admission for acute ischemic stroke
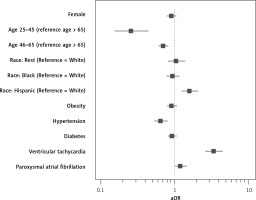
Several factors such as age > 66, Hispanic race (aOR = 1.622, 95% CI: 1.260–2.088, p < 0.01), presence of ventricular tachycardia (aOR = 3.426, 95% CI: 2.630–4.463, p < 0.01) and paroxysmal atrial fibrillation (aOR = 1.211, 95% CI: 1.017–1.442, p = 0.032) were associated with a higher risk of inpatient deaths among OSA patients with AIS. Meanwhile, hypertensive patients with OSA in the AIS group had a lower mortality risk (aOR = 0.649, 95% CI: 0.533–0.791, p < 0.01) (Table II, Figure 1). OSA patients were less likely to die (2.5%) than non-OSA groups (3.8%) when admitted for AIS (aOR = 0.738, 95% CI: 0.691–0.789, p < 0.01).
Table II
Possible factors influencing mortality among OSA patients admitted with AIS
Our study also identified 664,530 patients hospitalized for AMI. 8.9% of them had a diagnosis of OSA (Table III). The OSA AMI patients were more likely to be males (71.9% of OSA cases, and 10.2% of AMI males had OSA) and White (76.9% of OSA cases, 9.6% of Whites with AMI had OSA). While 52.3% of AMI patients with OSA were aged 66 or over, the incidence of OSA among those with AMI and aged 46-65 was highest (9.7%, p < 0.01). Compared to non-OSA patients with AMI, OSA patients with AMI were also more likely to be obese (51.5%, compared to 18.0%, p < 0.01), hypertensive (90.5%, compared to 82.1% < p < 0.01), and diabetic (56.5%, compared to 39.8%, p < 0.01). Statistically significant results were also found as OSA patients with AMI had a higher incidence of paroxysmal atrial fibrillation (13.8%, compared to 9.5%, p < 0.01) but a lower incidence of ventricular fibrillation (2.5%, compared to 3.3%, p < 0.01).
Table III
Characteristics of obstructive sleep apnea patients admitted for acute myocardial infarction
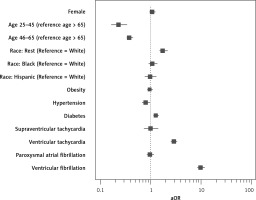
Moreover, OSA patients with AMI were more likely to die if they were aged 66 or over, not classified as “White, Black or Hispanic” (compared to Whites) (aOR = 1.771, 95% CI: 1.496–2.095, p < 0.01), diabetic (aOR = 1.276, 95% CI: 1.145–1.421, p < 0.01), or reported ventricular tachycardia (aOR = 2.937, 95% CI: 2.567–3.360, p < 0.01) or ventricular fibrillation (aOR = 9.847, 95% CI: 8.453–11.471, p < 0.01). A lower OR was calculated among those with hypertension (aOR = 0.800, 95% CI: 0.675–0.949, p < 0.010) (Table IV, Figure 2). 2.8% of OSA patients with AMI died, while 4.7% of patients without OSA and with AMI died (aOR = 0.672, 95% CI: 0.637–0.709, p < 0.01).
Table IV
Possible factors influencing mortality among OSA patients admitted with AMI
Discussion
Our study using the 2019 NIS confirmed that patients with OSA had a lower risk of dying when hospitalized with AMI (aOR = 0.672) or AIS (aOR = 0.738) in the United States. Similar results were also previously described by Mohananey et al. for outcomes of STEMI among OSA patients (aOR = 0.78) [3] and by Lapow et al. for patients admitted for AIS (aOR = 0.76) [6]. The lower mortality rate can be attributed to a possible higher quality of care, and more aggressive treatment protocols for patients who have already been previously diagnosed with OSA [3]. The higher incidence of OSA among men for both AMI and AIS can be narrowed down to the higher incidence of OSA in general in men compared to women [7–9].
We further identified a higher incidence of OSA among AIS and AMI patients in the age group of 46–65, which could be linked with the lifestyle of those adults, the increased mortality, and lower prevalence in the older groups [10]. Adults aged 66 or over showed a higher mortality rate than their younger counterparts due to the possible presence of more commodities predisposing them to various complications on admission for either AIS or AMI [11, 12]. While Whites presented with a higher incidence of OSA in both AMI and AIS, our study also showed that Hispanics with AIS had a higher mortality risk than Whites, while those classified as “not White, Black, or Hispanic” were more likely to die following admission for AMI if they also had OSA. Dudley et al. confirmed that the outcome of OSA is influenced by both genetic risk factors and the severity of OSA [13]. In our study, the HCUP database did not classify OSA in terms of severity, and we, therefore, encourage further studies based on a classification that could help better understand the results. Ramos et al. also previously reported that Hispanics were twice as likely to have a stroke and had several risk factors [14].
The relationship between obesity and OSA has been well established. Several studies have also shown that treating obesity and reducing weight can help improve the severity of OSA and the clinical outcomes of patients suffering from several conditions brought on by obesity and OSA [15]. While no statistically significant differences in mortality outcomes were found among OSA patients who were also obese, physicians are advised to encourage various health protocols among their OSA patients to improve their long-term survival. OSA is closely associated with diabetes and obesity. A higher incidence of diabetes was noted among OSA patients in both AMI and AIS groups. Since diabetics tend to have a higher BMI, they are more likely to develop OSA than non-obese groups, and up to 83% with type 2 DM may have undiagnosed OSA [16–19]. A higher risk of dying was also seen among diabetic OSA patients compared to non-diabetic OSA patients following admission for AMI, which agrees with past studies describing various pathways among people with diabetics which predispose them to a more severe outcome and further complications following an AMI [20–23].
A very high percentage of OSA patients with AMI and AIS had a history of hypertension in our study. Several studies have confirmed that OSA can provoke a rise in both systolic and diastolic pressures. While OSA episodes tend to occur at night, several patients have reported higher blood pressure even during the day [24]. Hypoxemia and hypercapnia during episodes of OSA can trigger a rise in sympathetic activation, leading to higher levels of catecholamine [25]. The lower mortality seen among hypertensive OSA patients than non-hypertensive OSA patients in both AIS and AMI groups in our study could be linked with the early prevention methods and blood pressure medications that the patients may have been on. The use of CPAP among OSA patients can help them reduce their blood pressure, which could also contribute to a better long-term outcome [26].
During an OSA episode, the patient usually experiences a low level of oxygen and a high level of carbon dioxide, which triggers several humoral and neuroendocrine feedback pathways to provoke several changes [27]. Gami et al. and Javaheri et al. both found a strong risk of atrial fibrillation in OSA patients, and similar results were obtained in our studies as OSA patients with AMI and AIS reported a higher incidence [28–30]. A higher mortality risk was also noted among the AIS cases in our study. We also noted a higher incidence of ventricular tachycardia in AIS patients, while the mortality in OSA patients with ventricular tachycardia was higher in both AIS and AMI groups. Lower incidence of ventricular fibrillation but higher mortality in OSA patients with ventricular fibrillation were seen among those with AMI. In a similar study, Salama et al. hypothesized that ventricular tachycardia may not be an essential factor for having ventricular fibrillation among OSA patients, but the causality is not fully understood [31]. Finally, a lower incidence of SVT was observed in our study among the AIS group, which could be linked to the reduction of SVT seen in OSA patients using CPAP [32].
While HCUP’s NIS allowed us to evaluate the patient characteristics and their outcomes following an AMI or AIS hospitalization, the study has several limitations. The database did not classify the severity of OSA in our sample group. Furthermore, we were unable to provide a follow-up for the patients who were discharged alive in the study, to rule out any post-discharge mortality or readmission leading to mortality.
In conclusion, while an overall lower mortality rate was seen among OSA patients following admission for AMI or AIS, the patients had several comorbidities that should be carefully managed to prevent a worse outcome.