Introduction
As a result of systematic increase in average life expectancy, there is a growing number of people who have a positive history of cardiovascular diseases, such as acute ischemic cerebrovascular events (AICEs). An AICE can occur in the form of a transient ischemic attack (TIA) or an ischemic stroke (IS) [1].
As the second death cause in the world and main reason for permanent invalidity and disability in the adult population, the stroke creates serious social and economic consequences on a global scale [2–4].
Many clinical trials have presented influence of insulin-like growth factor (IGF-1) on prognosis in IS [5–11]. This polypeptide, by activating receptors that are present in most tissues, including the brain, mediates the anabolic activity of growth hormone (GH) and its impact on growth and maturation processes, as well as organisms’ survival time [12]. It is produced in the liver and other organism’s tissues. Its particularly intensive synthesis is present in the developing brain [13, 14]. Experimental studies have shown neuroprotective activity of IGF-1, which directly modulates neuronal activity by inducing neurogenesis through progenitor cells proliferation and their maturation in neural direction [15]. It is also important in synaptogenesis and myelination, it stimulates angiogenesis as a response to hypoxia caused by brain ischemia or trauma. IGF-1 is an apoptosis inhibitor in the mechanism of protein kinase activation, it is an endogenous “survival factor” for neurons, glial and endothelial cells [13, 14, 16]. By stimulating differentiation of neural and oligodendrocyte progenitors, IGF-1 may encourage damaged nervous tissue regeneration [17, 18]. Through its anti-inflammatory effects, it probably benefits the evolution of the ischemic focus of the brain [19].
IGF-1 provides protection for the cardiovascular system in both endocrine and para endocrine mechanisms [20, 21].
Experimental studies have shown that IGF-1 administration reduces the stroke area and influences the improvement of neurological function disorders caused by cerebral ischemia [22].
Only a few studies have presented detailed data about IGF-1 influence on the course of the subacute phase of AICE.
The aim of the study was to assess the prognostic value of serum IGF-1 concentration in patients in the subacute phase of AICE and to relate serum IGF-1 level to neurological dysfunction.
Material and methods
The study included 86 participants.
The study group consisted of 56 patients (30 women, 26 men) aged between 53 and 97 years (mean age: 75.5 years), who were consecutively admitted to the Department of Neurology of the Faculty of Medicine and Dentistry of the Medical University of Warsaw on the first day of an AICE.
The control group consisted of 30 healthy volunteers (19 women, 11 men) aged between 50 and 72 years (mean age: 57.7 years).
Inclusion criteria and exclusion criteria of the study and control groups are presented in Tables I and II.
Table I
Inclusion and exclusion criteria of the study group
Table II
Inclusion and exclusion criteria of the control group
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Bioethical Commission of the Medical University of Warsaw (KB/56/2007).
Prior to study inclusion, patients and participants from the control group were informed about the course and aims of the study, as well as about the study scope and possible complications. They were informed about the possibility of waiving participation in the study at any time without giving a reason. Information was provided orally and in writing. Informed written consent was obtained from all study participants (or their legal guardian).
General and neurological examination, biochemistry tests, head CT or MRI scans, internal carotid and vertebral arteries ultrasound, ECG, echocardiography and chest X-ray were performed in the study group. Patients had IGF-1 serum concentration determined on the first day of the disease.
The control group underwent internal check-up, neurological examination and measurement of serum IGF-1 blood concentration.
IGF-1 serum concentration was determined with the chemiluminescence method, with the use of SIEMENS IMMULITE 2000 analyzer with analytic sensitivity of 20 µg/l. We collected 3 ml of blood from the ulnar vein on empty stomach to the plastic test-tube. The blood was centrifuged at a speed of 3500 per minute.
The diagnosis of IS or TIA was made according to the definitions on the basis of clinical symptoms and head imaging.
The course of AICE was assessed on the basis of neurological examination performed on the 1st and 9th day. The National Institute Health Stroke Scale (NIHSS) and modified Rankin Scale (mRS) were used. < 5 points in the NIHSS scale on the 9th day or improvement of at least 6 points in the NIHSS scale on the 9th day were considered as a positive course of AICE. Factors such as a marked neurological deficit on the 9th day (> 4 points in the NIHSS scale and/or > 2 points in mRS), no improvement or death during hospitalization defined a poor outcome.
We analyzed the correlation between IGF-1 concentration and the clinical condition of patients on days 1 and 9 of the onset of acute cerebral ischemia, the extent of ischemic lesion, risk factors and the course of the disease.
The following methods of statistical analysis were used to evaluate the studied variables:
One-dimensional analyses:
standard descriptive statistics were used to describe quantitative variables: mean values, standard deviations as well as medians and ranges,
the Shapiro-Wilk Test was used to verify the hypothesis that the distributions of the analyzed quantitative variables are consistent with the normal distribution,
when comparing two groups for quantitative variables, the non-parametric Wilcoxon test for independent samples (Wilcoxon Rank-Sum test) was used because the distributions of these variables showed deviations from the normal distribution,
when comparing several groups for quantitative variables, the Kruskal-Wallis test was used,
the analysis of correlation for quantitative variables was based on Spearman’s coefficients, the relationship between the course of acute cerebral ischemic events and the concentration of IGF-1 in the serum and other factors influencing the prognosis was examined. When assessing the strength of the relationship between the parameters, the following ranges of the correlation coefficient were used:
relationships between qualitative variables were tested in the contingency table layout using the χ2 test or Fisher’s Exact Test when the expected values in the table cells were not large enough.
Multivariate analyses
generalized regression models (GLM) were used:
– to investigate the influence of factors determining the course of acute cerebral ischemic events on the patient’s condition on the ninth day after AICE,
– to compare the IGF-1 concentration values in the study and control groups, taking into account the influence of age,
the selection of optimal models was based on AIC statistics (Akaike Information Criterion).
The p-value of 0.05 was adopted as a statistically significant level. The calculations were made in the SAS v. 14.1 System.
Results
Patients with AICE had lower IGF-1 concentrations in univariate analysis in comparison to the control group. The data are presented in Table III.
Table III
Analysis of the age distribution and IGF-1 [µg/l] serum concentration in patients and control group
| Parameter | N | Mean | Standard deviation | Median | Minimum | Maximum |
|---|---|---|---|---|---|---|
| Patients: | ||||||
| Age | 56 | 75.5 | 11.3 | 76.0 | 53.0 | 97.0 |
| IGF-1 | 56 | 104.7 | 53.0 | 104.4 | 9.7 | 261.0 |
| Control group: | ||||||
| Age | 30 | 57.5 | 5.4 | 56.0 | 50.0 | 72.0 |
| IGF-1 | 30 | 145.9 | 49.7 | 144.5 | 62.4 | 292.0 |
After exclusion of age influence on the IGF-1 levels, no statistically significant differences were present (p = 0.982).
There were statistically significant differences between patients with TIA and IS in IGF-1 ranges and initial neurological deficit assessed with NIHSS. The data are presented in Table IV.
Table IV
Comparison of IGF-1 [µg/l] blood serum concentration, age, severity of initial neurological syndrome (according to NIHSS) in patients with TIA (transient ischemic attack) and IS (ischemic stroke)
Patients with a mild neurological deficit (NIHSS < 5 points) had higher IGF-1 serum concentrations on the first day of AICE in comparison to patients with a higher baseline neurological deficit (NIHSS > 4 points). Medians were 114 µg/l and 85.5 µg/l, respectively (p = 0.049).
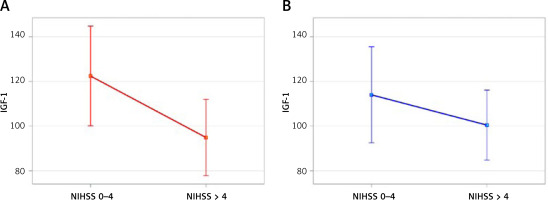
Taking into consideration age as a factor that significantly influences IGF-1 levels, we observed only the tendency for lower IGF-1 concentration in patients with higher severity of the neurological syndrome. The difference was, however, not statistically significant (p = 0.337) (Figure 1).
Figure 1
IGF-1 concentration and primary neurological deficit intensifiction (small neurololoical syndrome – NIHSS 0–4, relevant neurological syndrome – NIHSS > 4). A – accodrding to the model (p = 0.062), B – upon cerrection to the age factor (p = 0.337)
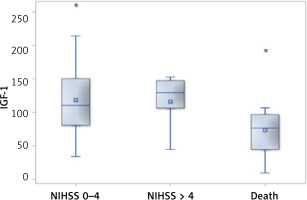
In patients with a milder neurological deficit found on the 9th day of the stroke, IGF-1 concentrations were significantly higher compared to those who died. The median IGF-1 values for these groups were 111 ng/ml and 76.9 ng/ml, respectively (p = 0.0059) (Figure 2).
In univariate analysis, patients with a favorable course of acute ischemic events presented higher IGF-1 concentrations in comparison to patients with a fatal course. Medians for both groups were 111 µg/l and 90.5 µg/l, respectively (p = 0.040).
In patients with a poor outcome, the severity of the primary neurological syndrome was a strong determining factor (p < 0.000). They were also older (p = 0.007), had higher glycemia (p = 0.017), leukocytosis (p = 0.006), elevated CRP (p < 0.000), lower fT3 (p = 0.016) and shortened APTT time (p = 0.015). Patients with a poor outcome of AICE course often experienced atrial fibrillation (p = 0.007), the mechanism of a cardioembolic stroke, stroke caused by large vessels disease and a stroke of undetermined etiology. The favorable outcome was the case in all patients with a lacunar stroke, while a poor final outcome concerned patients with a hemispheric stroke (p < 0.000).
There was a statistically significant negative correlation of IGF-1 concentration with the severity of the initial neurological syndrome evaluated in the NIHSS scale, with the severity of the clinical condition assessed on the 9th day in the modified Rankin scale, with CRP, fT3 and age. The data are presented in Table V.
Table V
Correlations of IGF-1 concentration with the studied biochemical and clinical parameters in patients with AICE
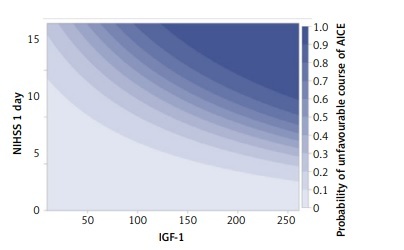
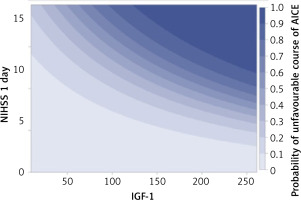
After considering in the multidimensional analyses of other strong factors determining the course of AICEs and their relationship with IGF-1, it was found that high IGF-1 values independently determined the worse course of AICE, and this relationship was most strongly expressed in patients with a significant neurological deficit present on the first day of AICE (Figure 3).
Discussion
There were no statistically significant differences in IGF-1 levels in multivariate analysis between the control group and the study group after exclusion of age influence. However analysis of our results showed serum IGF-1 level differences among patients with TIA and IS.
Some epidemiological studies noted an inverse relationship between IGF-1 plasma level and the risk of IS [23, 24]. However in the acute phase of stroke, IGF-1 shifts from peripheral circulation to CNS. It is caused by the blood-brain barrier damage. Due to this fact, IGF-1 concentration determined in the acute phase of stroke does not have to illustrate the level before the disease.
Results of clinical trials assessing the influence of IGF-1 concentration on treatment effects in the short-term and long-term observation are not unequivocal [5–7, 9, 11, 25].
Our study assessed the influence of IGF-1 concentration on the 1st day of IS or TIA on symptoms evolution only in the per acute phase of AICE – during the time when a patient needs an intensive medical supervision. There have been reports claiming that patient’s assessment in the first week of the ischemic stroke is a very sensitive test that allows to predict further prognosis. The usage of the NIHSS scale allows to state the intensification of the neurological deficit stage and to compare the patient’s condition in time [26–28].
Our study presented a statistically significant difference between IGF-1 levels in patients with TIA and IS – the medians were 123.6 µg/l and 96.9 µg/l, respectively (p = 0.044).
We noted that patients with a more severe neurological deficit had lower IGF-1 levels in comparison to patients with mild intensification of the neurological syndrome. There was, however, a higher average age in the group of patients with a worse neurological condition. It is widely known that IGF-1 drops physiologically with age, which was also illustrated in this study (p < 0.001). Elimination of this factor in multivariate analysis showed the tendency for lower IGF-1 concentration in patients with a more severe stroke-related neurologic deficit (p = 0.337).
Dong et al. claimed that a low IGF-1 concentration is significantly connected with the severity of the stroke measured with the NIHSS scale [25].
Differences in IGF-1 serum concentrations between patients with mild and severe neurological disability may be explained with organism’s reaction to acute brain injury, which starts the mechanisms causing IGF-1 shift from peripheral circulation to CNS. IGF-1 transfers from serum to the brain tissue through the blood-brain barrier, which is disrupted throughout the first few weeks after AICE [18, 29, 30]. As a result of this process, serum IGF-1 concentration drops, which can be an indicator of its higher level in the brain.
Experimental studies, which showed the increase in IGF-1 expression and its receptors in damaged nervous tissue after acute brain ischemia, may present explanations for our findings [31, 32]. Moreover, other authors claim that the larger area of ischemic lesion is connected with the decreased IGF-1 serum concentration [33].
It is known that as a result of IS, IGF-1 crosses the blood-brain barrier [34]. It was proven by peripheral administration of radiolabeled IGF-1 just after inducing middle cerebral artery occlusion in rats. 30 min after IGF-1 administration, it was established that most of IGF-1 was bound with the stroke affected cerebral hemisphere neurons [35].
As neuroprotective and neurorepair IGF-1 effect has been repeatedly proven, low serum concentrations of IGF-1 in patients with a better neurological improvement in AICE may be due to mechanisms that cause IGF-1 to shift from the blood to the CNS [36].
Other clinical trials have also shown the decrease in IGF-1 serum concentration in ischemic stroke [25]. Schwab et al. stated that plasma levels of IGF-1 decreases in patients with the acute phase of ischemic stroke and the decrease is proportional to the extent of ischemic lesion. It was explained with post ischemic stroke need for growth factors, their changed tissue distribution or central inhibition of somatotropic axis [37]. Moreover decline in serum IGF-1 concentration happens in critical illness that causes massive activation of the immune system [38].
We are not able to supra vitally measure IGF-1 in brain tissue among people. However, the conclusions from experimental and clinical trials are important for the interpretation of the current study, as IGF-1 serum decrease may be an indicator of its increase in the brain.
Studies assessing changes and relations of IGF-1 in serum and in the cerebrospinal fluid in patients with the acute phase of ischemic stroke might contribute a lot. They have, however, restrictions due to the risk connected with a lumbar puncture in the acute phase of severe ischemic stroke.
The answer whether the changes of IGF-1 concentrations are a neuroprotective reaction to acute trauma or are the result of a high level from the time before disease is hard to establish and it requires further examinations.
Comparing the serum IGF-1 concentration in patients with a different course of AICE in a in univariate analysis, higher levels of IGF-1 were found in patients with a favorable course of AICE than in patients with a poor outcome (p = 0.040).
A clearly marked relation between a higher IGF-1 concentration and favorable AICE course was not proven in multivariate analysis, which included other factors affecting the course of AICE, such as primary intensification of the neurological syndrome or age. It turned out that high IGF-1 serum levels determined the worse course of AICE and relation between them was directly proportional to the neurological deficit on the first day of the disease.
A negative correlation between the initial neurological dysfunction (NIHSS 1 day) and serum IGF-1 indicates that the higher the NIHSS value, the lower the IGF-1 values. The neurological assessment according to the NIHSS scale on the 1st day is a very strong factor determining the patient’s final state (after 9 days) – the higher the NIHSS value on the 1st day, the greater the probability of a poor end state. As patients with high NIHSS scores on day 1 often have lower IGF-1 values, the univariate analysis of the effect of IGF-1 on the final state shows that lower IGF-1 values increase the risk of a poor course. However, this is a masked influence of a much stronger factor, which is the NIHSS value on the 1st day.
With a negative correlation between IGF-1 concentration and severity of primary neurological deficit assessed in the NIHSS scale and strong influence of the poor neurological condition (high NIHSS scale value) on AICE course, univariate analysis which did not take into account this relationship, might not have illustrated the real IGF-1 influence for outcome. Taking it into account, it turned out that higher IGF-1 serum concentrations increased the probability of a poor acute ischemic event course and the poorest prognosis was in patients with a serious primary condition corresponding with high IGF-1 levels. We might say that the observed negative correlation between a large neurological deficit on the first day and IGF-1 serum concentration was beneficial for patients, as high NIHSS values on the first day was accompanied with a low IGF-1 level.
Results of clinical trials assessing the influence of IGF-1 concentration on treatment effects in short-term and long-term observation are not unequivocal.
Denti et al. found the negative correlation between IGF-1 serum concentration and patients’ condition after a month, 3 months and 6 months after IS [7]. In another trial assessing the prognostic value of IGF-1 concentration in IS, Tang et al. reported on an important connection between a low IGF-1serum level on the first day of stroke and a poor final result, meaning death or poor functional status after 90 days in the mRS scale [10].
Opposed conclusions – a positive link between high IGF-1 serum levels in the acute phase of ischemic stroke and a poor result assessed 3 months after the stroke – were drawn in Armbrust et al. trial that included 404 patients [8].
Conclusions similar to ours were mentioned by Mattlage et al. They obtained a positive correlation between low IGF-1 concentrations in the first week of IS and a positive outcome in the first month after the stroke [11].
IGF-1 is a stable factor that can be easily measured and may be one of the stroke markers that may allow to predict its evolution. In the future, it may influence therapeutic decisions in patients who are at risk of severe disability or death. IGF-1 systemic treatment may be beneficial as the blood-brain barrier is damaged for a few weeks after cerebral ischemia [18]. It is possible to consider introducing IGF-1 directly to the brain via intra-arterial administration and IGF-1 treatment as additional to thrombectomy.
Our study suggests that patients with a large neurological deficit on the first day of the disease and with a high IGF-1 concentration require special clinical observation and probably more radical treatment as the prognoses among this group are the most serious.
The findings may have important implications for establishing AICE prognoses and they lead to further questions which should be foundations for future studies.
In conclusion, a high serum level of IGF-1 on the first day of AICE is an independent, predictive factor determining the unfavorable course of the subacute phase of stroke, and this relationship is proportional to the severity of the baseline neurological deficit.
There is a positive correlation between lowered IGF-1 serum concentration measured on the first day of AICE and the severity of the neurological deficit.