Introduction
The scientific community has been increasingly interested in the potential lipid-lowering effect of functional foods and nutraceuticals, as well as their combined use in the prevention of cardiovascular disease (CVD) [1]. In the last decade, red yeast rice and berberine were clearly the most investigated lipid-lowering nutraceuticals [2], being able to reversibly inhibit liver 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase and proprotein convertase subtilisin/kexin type 9 (PCSK9) [3].
In recent times, the efficacy of products containing the most potent lipid-lowering nutraceutical, red yeast rice, has been reduced by the application of a rule of the European Commission that quantitatively limits the daily use of monacolins in red yeast rice containing products to less than 2.9 mg [4]. A meta-analysis of 12 randomized double-blind, placebo controlled clinical trials including 1050 subjects concluded that berberine 500 mg associated with low-dosed red yeast rice is able to significantly reduce total cholesterol (TC; MD = –25.07 mg/dl, p < 0.001), triglycerides (TG; MD = –11.47 mg/dl, p < 0.001), low-density lipoprotein cholesterol (LDL-C; MD = –26.67 mg/dl, p < 0.001), and high-sensitivity C-reactive protein (hs-CRP: MD = –0.61 mg/l, p = 0.022), and to increase high-density lipoprotein cholesterol (HDL-C; MD = 1.84 mg/dl, p < 0.001) [5]. In this context, we clinically tested a new combined formulation, Zeta Colest, including berberine, low-dosed red yeast rice, but also milk thistle and guggul dry extract.
Material and methods
We conducted a double-blind, placebo-controlled, randomized, clinical study, whose objective was to assess the efficacy and safety profile of the new Zeta Colest nutraceutical formulation over a period of 8 weeks in patients at low risk for CVD and with elevated levels of LDL-C.
The study fully complied with the ethical guidelines of the Declaration of Helsinki and with the International Council for Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use Harmonized Tripartite Guideline for Good Clinical Practice. The study protocol was approved by the Ethical Committee of the University of Bologna (ZC20172201). All volunteers signed written informed consent to participate and were free to withdraw from the study at any time.
This study involved a sample of Italian free-living subjects with polygenic hypercholesterolemia recruited from the Cardiovascular Medicine Unit of the S. Orsola Malpighi University Hospital, Bologna, Italy.
Participants were eligible for enrolment if they were aged 30–70 years, with moderately high levels of LDL-C (LDL-C > 130 mg/dl and < 190 mg/dl) and an estimated 10-year cardiovascular risk < 5% according to the SCORE (Systematic COronary Risk Evaluation) risk charts, not requiring lipid-lowering treatments [6]. Exclusion criteria included TG > 400 mg/dl, uncontrolled hypertension, previous history of CVD, obesity (defined as body mass index (BMI) > 30 kg/m2), diabetes mellitus, uncontrolled thyroid diseases, use of medication or nutritional supplement that altered serum lipids (e.g. statins, ezetimibe, fibrates, omega-3 fatty acids and bile acid resins), alcoholism, pregnancy and breastfeeding.
All the patients were adhering to a low-fat Mediterranean diet for 4 weeks before randomization. They were also strongly recommended to avoid excessive intake of dairy and red meat derived products, in order to maintain an overall balanced diet. A specialist physician advised them to obtain around 50% of calories from carbohydrates, 30% from fat (6% saturated), and 20% from proteins, with a maximum cholesterol content of 200 mg/day and 35 g/day of fiber.
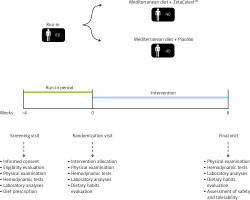
Randomization was stratified according to sex and age, with patients assigned (in a 1 : 1 ratio) to receive either Zeta Colest or keep following the diet previously detailed. The intervention period lasted 8 weeks. Before and at the end of the study, all patients were evaluated for clinical status, and physical, hemodynamic and laboratory assessments. The study timeline is reported in Figure 1.
The new Zeta Colest nutraceutical formulation used in the study contained dry extracts of Berberis aristata, red yeast rice, milk thistle and guggul (Table I).
Table I
Quantitative composition of the dietary supplement Zeta Colest tested in the clinical study
| Active ingredients | Quantity per pill [mg] |
|---|---|
| Berberis aristata d.e. Berberine | 400 340 |
| Red yeast rice extract Total monacolins | 98 2.9 |
| Milk thistle d.e. Silymarin | 87.5 70 |
| Guggul d.e. guggulsterones | 50 1.3 |
The dietary supplement and the placebo were manufactured and packaged by Erbozeta S.p.A. (San Marino), in accordance with Quality Management System ISO 9001:2008 and the European Good Manufacturing Practices (GMP), satisfying the requirements in the Code Of Federal Regulation title 21,volume 2, part 111.
On day 0, patients randomized to receive Zeta Colest or an undistinguishable placebo were provided with 2 boxes each containing 30 pills, and were instructed to take a pill once daily in the evening, for the entire duration of the study. All unused pills were retrieved for inventory during the end-of-study visit, and participants’ compliance was assessed by counting the number of returned pills.
Information gathered in the patients’ history included presence of atherosclerotic cardiovascular disease (ASCVD) and other systemic diseases, allergies and medications. Validated semi-quantitative questionnaires were used to assess demographic variables, recreational physical activity and smoking habits.
Dietary intake was estimated by 4-day records [7]. Analysis of diet composition was performed using the MètaDieta software (INRAN/IEO 2008 revision/ADI). Data were handled in compliance with the company procedure IOA87.
Waist circumference (WC) was measured in a horizontal plane at the end of a normal expiration, at the midpoint between the inferior margin of the last rib and the superior iliac crest. Height and weight were respectively measured to the nearest 0.1 cm and 0.1 kg, with subjects standing erect with eyes directed straight, wearing light clothes and with bare feet. BMI was calculated as body weight in kilograms divided by height squared in meters (kg/m2).
Blood pressure (BP) was measured in accordance with the recommendations of the International Guidelines for the management of arterial hypertension [8]. Resting systolic (SBP) and diastolic BP (DBP) were measured with a validated oscillometric device and a cuff of the appropriate size applied on the right upper arm. To improve detection accuracy, three BP readings were sequentially obtained at 2-minute intervals. The first reading was discarded, and the average between the second and the third reading was recorded as a study variable. Pulse pressure (PP) was calculated as the difference between SBP and DBP.
Following the current guidelines [9], during the clinical study endothelial function was evaluated through Endocheck (BC Biomedical Laboratories Ltd., Vancouver, BC, Canada), a method embedded within the Vicorder device that guarantees very good intra- and inter-operator reliability [10]. The measurement was carried out with patients in a supine position and in abstinence from cigarette smoking and caffeinated beverages for at least 12 h. After a 10-minute rest, the brachial pulse volume (PV) waveforms were recorded at baseline for 10 s and during reactive hyperemia. The BP cuff was inflated to 200 mm Hg for 5 min and PV waveforms were recorded for 3 min after the cuff was released. Endothelial reactivity (ER) was calculated as change in the PV waveform area, comparing waveforms before and during hyperemia through the equation √PV2/PV1, where PV1 represents PV at the baseline and PV2 represents PV during hyperemia [10].
The biochemical analyses were carried out on venous blood withdrawn early in the morning from the basilic vein. Patients were fasted for at least 12 h at the time of sampling. All of the laboratory analyses were centrally performed in the laboratory of our department, by trained personnel and in accordance with standardized methods described in detail elsewhere [11]. The following parameters were obtained or calculated through the appropriate formula: TC, TG, HDL-C, non-HDL cholesterol (non-HDL-C), LDL-C, apolipoprotein B-100 (Apo B-100), apolipoprotein A-I (Apo A-I), fasting plasma glucose (FPG), hs-CRP, γ-glutamyl transferase (γ-GT), alanine transaminase (ALT), aspartate transaminase (AST) and creatinine phosphokinase (CPK).
Safety and tolerability were evaluated during the study in order to detect any adverse event, clinical safety, laboratory findings, vital sign measurements, and physical examinations [12].
Statistical analysis
Data were analyzed using intention to treat by means of SPSS version 25.0 for Windows. Under the assumption of a 7% dropout rate, a sample of 40 patients/group was needed to provide more than 90% power to detect a 15% lower level of LDL-C in the actively treated group than in the control group with a 2-sided significance level of 0.05.
Baseline parameters with normal distribution were compared using Student’s t test. Between-group differences were assessed by the analysis of variance (ANOVA) followed by Tukey’s post-hoc test. All data were expressed as means and related standard deviations (SD). All tests were 2-sided. A p level of < 0.05 was considered significant for all tests.
Results
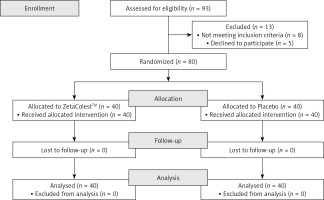
Overall, 93 patients were screened, and 80 (men: 49; women: 31) were randomized. All of them successfully completed the study according to its design. Moreover, no patient experienced any subjective or laboratory adverse event (dropout rate = 0%) and the compliance to the treatment was ~100% in both study groups (Figure 2).
Enrolled individuals maintained similar dietary habits from the randomization until the end-of-study visit, without significant changes in total energy, salt intake and coffee and alcohol consumption.
At baseline, clinical features and laboratory analyses were similar between groups (Tables II and III).
Table II
Clinical characteristics of the enrolled subjects at pre-run-in visit, baseline and after treatment in the actively treated group and in the control group
| Parameter | Pre run-in | Mediterranean diet + Zeta Colest (N = 40) | Mediterranean diet + Placebo (N = 40) | ||
|---|---|---|---|---|---|
| Baseline | Post-treatment | Baseline | Post-treatment | ||
| Age [years] | 52.9 ±9.1 | 52.3 ±8.5 | 52.8 ±9.6 | ||
| Height [m] | 1.69 ±0.12 | 1.69 ±0.10 | 1.70 ±0.12 | ||
| Weight [kg] | 73.1 ±13.9 | 72.3 ±11.1 | 72.0 ±9.1 | 73.2 ±13.7 | 71.9 ±12.5 |
| Body mass index [kg/m2] | 25.8 ±2.2 | 25.6 ±2.2 | 25.5 ±2.1 | 25.7 ±2.1 | 25.4 ±2.0 |
| Waist circumference [cm] | 94.2 ±9.7 | 92.9 ±9.5 | 92.5 ±9.4 | 93.7 ±9.1 | 92.6 ±8.7 |
| Heart rate [bpm] | 73 ±9 | 70 ±8 | 71 ±5 | 72 ±7 | 70 ±9 |
| Systolic blood pressure [mm Hg] | 124 ±9 | 124 ±8 | 123 ±6 | 124 ±7 | 122 ±9 |
| Diastolic blood pressure [mm Hg] | 78 ±7 | 77 ±5 | 77 ±4 | 78 ±6 | 77 ±5 |
| Endothelial reactivity | – | 1.35 ±0.21 | 1.39 ±0.15* | 1.33 ±0.22 | 1.36 ±0.23 |
Table III
Laboratory parameters of the enrolled subjects at pre-run-in visit, baseline and after treatment in the actively treated group and in the control group
| Parameter | Pre run-in | Mediterranean diet + Zeta Colest (N = 40) | Mediterranean diet + Placebo (N = 40) | ||
|---|---|---|---|---|---|
| Baseline | 8-week follow-up | Baseline | 8-week follow-up | ||
| Total cholesterol [mg/dl] | 233.6 ±14.1 | 229.7 ±12.9 | 194.8 ±12.3*° | 232.5 ±11.6 | 235.6 ±13.9 |
| LDL cholesterol [mg/dl] | 158.5 ±10.3 | 155.3 ±9.9 | 127.2 ±12.3*° | 161.1 ±11.3 | 163.8 ±12.3 |
| HDL cholesterol [mg/dl] | 51.1 ±3.4 | 51.4 ±2.5 | 52.3 ±2.7 | 49.8 ±3.3 | 50.1 ±2.9 |
| Non-HDL cholesterol [mg/dl] | 182.5 ±10.7 | 178.3 ±10.1 | 147.6 ±9.6*° | 183.7 ±10.1 | 187.4 ±9.3 |
| Triglycerides [mg/dl] | 119.8 ±26.8 | 114.8 ±25.2 | 103.6 ±23.9 | 110.7 ±26.3 | 119.6 ±21.7 |
| Apolipoprotein B [mg/dl] | 111.2 ±13.1 | 106.7 ±10.1 | 88.6 ±11.4*° | 110.1 ±9.5 | 111.5 ±10.4 |
| Apolipoprotein AI [mg/dl] | 152.6 ±16.5 | 154.2 ±16.2 | 154.4 ±14.4 | 160.4 ±17.6 | 155.9 ±18.3 |
| Fasting plasma glucose [mg/dl] | 89.9 ±7.6 | 89.1 ±7.4 | 88.5 ±6.7 | 87.8 ±6.5 | 86.7 ±7.2 |
| Alanine aminotransferase [U/l] | 24.1 ±4.6 | 22.5 ±4.1 | 26.3 ±3.6 | 23.4 ±4.3 | 26.2 ±4.1 |
| Aspartate aminotransferase [U/l] | 20.9 ±3.4 | 21.8 ±3.6 | 23.6 ±3.4 | 21.7 ±3.1 | 21.2 ±3.5 |
| γ-glutamyl transferase [U/l] | 22.8 ±7.5 | 22.1 ±7.5 | 22.9 ±8.4 | 21.4 ±7.1 | 24.4 ±8.8 |
| Creatine phosphokinase [U/l] | 144 ±33 | 130 ±55 | 145 ±71 | 141 ±67 | 154 ±63 |
| hsCRP [mg/dl] | 2.96 ±0.19 | 2.98 ±0.24 | 2.87 ±0.17*° | 2.76 ±0.21 | 2.71 ±0.23 |
During the study, we did not observe any significant change in the anthropometric measurements, BP, HDL-C, FPG, liver parameters, and CPK (p > 0.05 in both groups).
No statistically significant difference was detected between the treatment groups, both at the baseline and after active treatment, except endothelial reactivity, which significantly improved versus baseline (but not versus placebo) in the group treated with the tested combined nutraceutical.
Total, non-HDL, LDL-cholesterol and apolipoprotein B100 plasma levels significantly improved both after active treatment versus baseline (all, p < 0.05) and diet-only treated group (all, p < 0.05). In particular, percentage LDL-C change vs. baseline was –18.1 ±1.9% (p < 0.05).
No significant change was observed in the diet only treated group as regards all investigated metabolic parameters.
Discussion
An early and long-term reduction of LDL-C plasma level seems to be prognostically relevant in subjects with mild-to-moderate hypercholesterolemia in primary prevention for CV events [13], even though a cost-benefit analysis of drug use has not yet been undertaken [14].
In this randomized, diet-controlled, clinical study, the short-term supplementation with a combined lipid-lowering nutraceutical was shown to be able to significantly reduce plasma levels of TC (–15.2 ±1.4%), LDL-C (–18.1 ±1.9%), non-HDL-C (–17.7 ±1.8%), Apo-B (–16.9 ±1.9%) and hs-CRP (–3.7 ±0.7%) vs. baseline.
These effects are larger than the ones we observed with the association of berberine and red yeast rice at mildly higher dosages [5]. The reason is probably related to the other bioactive components included in Zeta Colest. In particular, silymarin seems to increase the berberine lipid-lowering efficacy by increasing its bioavailability [15], beyond a mild direct LDL-lowering effect of silymarin per se [16]. On the other hand, guggulsterones could also exert some LDL-lowering effect by inhibiting CYP7A [17], having already been demonstrated to exert an additive effect on that of red yeast rice in a small randomized clinical trial [18].
Endothelial reactivity also mildly but significantly improved by 2.96 ±0.23% with Zeta Colest: this pulse volume displacement was small, but available data show that even small improvements in endothelial reactivity are associated with a significant reduction in cardiovascular disease risk [19].
Our study has some limitations. The main one is related to the relatively short length of the study, although it was sufficient to observe metabolic and vascular improvements in the actively treated group. Furthermore, the tested supplement contained a number a bioactive components, so it is difficult to quantify the contribution of each of them to the whole final observed effect. Definitely, the current study is preliminary and further research is needed to more deeply investigate the long-term effect of bergamot extract on a broader range of parameters. However, the tested combined nutraceutical has been shown to improve a relatively large number of cardiometabolic risk factors, in agreement with what is expected for a well-designed combined nutraceutical [20]. Considering the recent European Commission concerns about the use of red yeast rice products containing high dosages of monacolin K [21, 22], the current development of new combined nutraceuticals is needed to balance the strong reduction in the daily dose of monacolins required by the new EU rules.
In conclusion, in the short term, the tested combined nutraceutical was well tolerated and significantly improved lipid metabolism, systemic inflammation and vascular function in overweight subjects with suboptimal cholesterolemia.