Introduction
Coronary artery bypass grafting (CABG) is a key treatment for advanced coronary artery disease (CAD), especially in patients with multivessel disease or when percutaneous coronary intervention (PCI) is not an option [1]. While CABG is widely used and significantly enhances long-term survival [2, 3], it can lead to certain complications. One major concern is graft failure, which occurs in 10% to 50% of cases over time [4–7]. This is mainly caused by thrombosis and intimal hyperplasia. To address this, antiplatelet therapy plays a crucial role in improving graft patency and overall patient outcomes.
Aspirin has long been the gold standard for antiplatelet therapy in post-CABG patients due to its proven efficacy in reducing graft occlusion and cardiovascular events [8, 9]. However, recent evidence has increasingly supported the use of dual antiplatelet therapy (DAPT) with a P2Y12 inhibitor in addition to aspirin, particularly in patients at high risk of thrombotic events [10]. DAPT, while more effective in reducing the incidence of major adverse cardiovascular and cerebrovascular events (MACCE), also carries an increased risk of bleeding, which can complicate postoperative recovery and may demand additional surgical interventions [11, 12].
The choice of P2Y12 inhibitor in DAPT is a critical decision. Clopidogrel, a thienopyridine that irreversibly inhibits the P2Y12 receptor, has been widely used due to its relatively favorable bleeding profile and extensive clinical experience. However, it is a prodrug requiring hepatic activation, and genetic polymorphisms affecting CYP2C19 can result in variable antiplatelet effects, leaving up to 30% of patients at risk of inadequate platelet inhibition [13]. This has led to the increasing use of newer, more potent P2Y12 inhibitors, such as ticagrelor, which do not require metabolic activation and offer more consistent platelet inhibition [14].
Ticagrelor, a direct-acting reversible P2Y12 inhibitor, has been shown to reduce the risk of MACCE more effectively than clopidogrel, particularly in acute coronary syndrome (ACS) settings [15]. However, its association with increased bleeding, especially in the context of surgery, poses significant challenges in its perioperative management [16]. Prasugrel, another potent irreversible inhibitor of the P2Y12 receptor, has demonstrated superior efficacy to clopidogrel in preventing thrombotic events, albeit with a higher risk of major bleeding [17].
Although dual antiplatelet therapy (DAPT) offers clear benefits, important questions remain about the best choice of P2Y12 inhibitor and the ideal timing to stop these medications before CABG. These decisions aim to balance reducing bleeding risks while maintaining effective antithrombotic protection [18]. Current guidelines advise against starting prasugrel in patients needing CABG due to its higher bleeding risk. However, prasugrel has not been thoroughly studied in this setting, creating a gap in recommendations for its use around the time of surgery [19]. Similarly, while ticagrelor has not been shown to significantly increase CABG-related bleeding compared to clopidogrel, it is linked to a higher overall risk of major bleeding. This adds complexity to its management during the perioperative period. The limited data on both agents in the context of CABG contributes to ongoing uncertainty about their optimal role in dual antiplatelet therapy for these patients [19].
This systematic review and meta-analysis aim to address these critical gaps by evaluating the comparative efficacy and safety of ticagrelor, prasugrel, and clopidogrel in patients undergoing CABG. By analyzing data from around 10,000 patients, we seek to provide evidence-based recommendations on the optimal antiplatelet strategy for CABG, with the goal of improving postoperative outcomes while minimizing the risk of adverse events.
Methodology
Data source, search strategy, and eligibility criteria
This systematic review and meta-analysis were conducted in strict adherence to the Cochrane Handbook for Systematic Reviews of Interventions and the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [20, 21]. The study protocol was pre-registered on the International Prospective Register of Systematic Reviews (PROSPERO) under ID 545557. Comprehensive searches were performed across electronic databases including MEDLINE, Scopus, and the Cochrane Central Register of Controlled Trials, covering the period from their inception to September 30, 2024. A detailed search strategy, including specific keywords and MeSH terms, is available in the Supplementary Tables SI.
To identify relevant articles, reference lists of obtained trials, review articles, and previous meta-analyses were manually screened. All articles retrieved after the systematic search were exported to Zotero (Version 6.0, Corporation for Digital Scholarship, Vienna, VA, USA), where duplicates were identified and removed. Studies were included if they met the following criteria: (i) CABG for coronary heart disease in patients ≥ 18 years old with at least two antiplatelet arms (clopidogrel vs. ticagrelor or prasugrel); (ii) clopidogrel + aspirin as the control group; (iii) outcome reporting as patient count or percentage; (iv) at least one of the following outcomes: all-cause mortality, major bleeding, re-bleeding, myocardial infarction, stroke, RBC transfusion, or platelet transfusion; (v) original articles, including RCTs, case-control, and cohort studies. Any review articles, ongoing RCTs, and studies conducted in a language other than English were excluded.
Data extraction and quality assessment
Two independent reviewers (JA and MA) initially assessed the remaining articles based on title and abstract, followed by a full-text screening to determine relevance to the eligibility criteria. Any discrepancies that arose were resolved by a third reviewer (SW).
Trial characteristics, baseline demographics, outcomes, and safety data were extracted onto a predesigned Excel spreadsheet. The quality assessment of the identified articles was conducted by two reviewers using the Cochrane Collaboration’s risk of bias tool for RCTs [22], and the Newcastle-Ottawa scale was utilized to evaluate the quality of observational studies [23]. Evaluation criteria were based on comparability, selection, and outcome or exposure of the included studies (Supplementary Table SII and SIII).
Statistical analysis
We reported outcomes as odds ratios (ORs) with 95% confidence intervals (CIs) and pooled them using the Der Simonian-Laird random-effects model [24]. We used forest plots to visualize pooled results and funnel plots to assess publication bias. Heterogeneity across trials was assessed using Higgins I2. The χ2 test was performed to evaluate differences between the subgroups. We assessed publication bias using Egger’s regression test for outcomes with 10 or more studies.
We conducted a sub-group analysis to determine whether the following factors influenced the effect size: (i) study design (RCT vs. observational) and (ii) instance of drug administered (before CABG vs. after CABG). Furthermore, we performed a sensitivity analysis by excluding studies that appeared as outliers in the forest plot. A two-tailed p < 0.05 was considered statistically significant. All statistical analyses were conducted using R (Version 4.4.1; R Foundation for Statistical Computing, Vienna, Austria).
Results
Aspirin plus ticagrelor versus aspirin plus clopidogrel
The PRISMA flowchart (Supplementary Figure S1) presents a concise overview of the search and trials section. Seventeen trials were included, comprising two RCTs and fifteen observational studies, which assessed the effect of aspirin plus ticagrelor versus aspirin plus clopidogrel on patients undergoing CABG and their postoperative outcomes [24–41]. Most of the included studies were found to have a low risk for bias, except for Dery and Tang, which had a moderate risk [26, 34]. A total of 8,891 patients participated in these trials (n = 4,148 in the aspirin + ticagrelor arm and n = 4,743 in the aspirin + clopidogrel arm). The mean age of the patients was 65.9 years, with males comprising 75.6% of the population. Table I summarizes the baseline characteristics of patients in each included trial.
Table I
Summary of baseline characteristics in studies comparing ticagrelor and clopidogrel effects in patients undergoing coronary artery bypass graft
Major bleeding
Fifteen studies reported major bleeding. Meta-analysis of these studies showed no significant difference between the drugs in preventing major bleeding (OR = 1.01 [0.78, 1.31]; p = 0.92, I2 = 52%; Figure 1 A). The funnel plot showed a symmetrical distribution, suggesting robust results (Supplementary Figure S2). Egger’s regression test showed no evidence of publication bias (p = 0.10). Subgroup analysis of ticagrelor discontinuation for within 3 days versus more than 3 days was possible for eight studies, with six of them providing us with data for both discontinuation groups. The results of this analysis were statistically significant, favoring ticagrelor being discontinued more than 3 days prior to surgery (OR = 0.62 [0.47, 0.83]; p < 0.01, I2 = 45%; Figure 1 B). Conversely, the analysis favored clopidogrel when discontinuation occurred less than 3 days prior to surgery (OR = 1.57 [1.04, 2.36]; p < 0.01, I2 = 63%; Figure 1 B). Other subgroup analyses did not demonstrate any effect modification by the instance of drug administered (P-interaction: 0.99; Supplementary Figure S3) or by study design (P-interaction: 0.39; Supplementary Figure S4). Excluding Varma and Voetsch during the sensitivity analysis reduced the heterogeneity and effect size (OR = 0.86 [0.71, 1.05]; p = 0.14, I2 = 21%; Supplementary Figure S5).
Figure 1
The effect of ticagrelor on (A) major bleeding, (B) major bleeding by ticagrelor discontinuation timing (C) re-bleeding, (D) platelet transfusion, and (E) all-cause mortality in CABG patients. Forest plots show the effect of ticagrelor versus clopidogrel, in combination with aspirin on outcomes in patients undergoing coronary artery bypass graft surgery. Ticagrelor significantly reduced major bleeding when discontinued more than 3 days before surgery, while clopidogrel was more effective if ticagrelor was discontinued less than 3 days prior to surgery. No significant differences were observed in the overall incidence of major bleeding, re-bleeding, platelet transfusions, or all-cause mortality

Re-bleeding
Re-bleeding was reported in twelve studies. The meta-analysis of these studies revealed that ticagrelor trended towards preventing re-bleeding (OR = 0.85 [0.68, 1.08]; p = 0.18, I2 = 3%; Figure 1 C), although the P-value indicates that this result is not statistically significant. The funnel plot revealed a symmetrical distribution, suggesting robust results (Supplementary Figure S6). Egger’s regression test showed no evidence of publication bias (p = 0.95). Subgroup analysis did not demonstrate any effect modification by instance of drug administered (P-interaction: 0.30; Supplementary Figure S7) and by study design (P-interaction: 0.22; Supplementary Figure S8). Sensitivity analysis by removing Held and Varma reduced heterogeneity and resulted in a statistically significant effect size (OR = 0.78 [0.61, 0.99]; p = 0.04, I2 = 0%; Supplementary Figure S9).
Platelet transfusion
Platelet transfusions right after CABG were reported in seven studies. Meta-analysis of these studies showed that neither drug was superior in reducing the need for platelet transfusion (OR = 0.89 [0.73, 1.08]; p = 0.23, I2 = 42%; Figure 1 D). The funnel plot revealed symmetrical distribution, signifying robust findings (Supplementary Figure S10). Sensitivity analysis by removing Voetsch and Dery reduced the heterogeneity and resulted in a statistically significant effect size (OR = 0.83 [0.69, 0.99]; p = 0.04, I2 = 31%; Supplementary Figure S11).
All-cause mortality
Ten studies reported all-cause mortality. Meta-analysis of these studies showed no significant difference between the drugs in preventing all-cause mortality (OR = 1.04 [0.65, 1.65]; p = 0.88, I2 = 54%; Figure 1 E). The funnel plot showed a symmetrical distribution, indicating robust results (Supplementary Figure S12). Egger’s regression test showed no evidence of publication bias (p = 0.14). Subgroup analysis showed a statistically significant reduction in all-cause mortality with post-CABG ticagrelor (OR = 0.49 [0.33, 0.73]; p < 0.01, I2 = 0%; Supplementary Figure S13). Sensitivity analysis by removing Held reduced the heterogeneity and increased the effect size (OR = 1.22 [0.83, 1.79]; p = 0.32, I2 = 14%; Supplementary Figure S14).
RBC transfusion
RBC transfusions right after CABG were reported in nine studies. Meta-analysis of these studies showed no significant difference between the drugs in reducing the need for RBC transfusion (OR = 1.02 [0.89, 1.17]; p = 0.80, I2 = 12%; Supplementary Figure S15). The funnel plot presented a largely symmetrical distribution, suggesting robust results (Supplementary Figure S16). Sensitivity analysis by removing Wang reduced the heterogeneity (OR = 1.00 [0.88, 1.13]; p = 0.98, I2 = 0%; Supplementary Figure S17).
Stroke
Stroke was reported in eight studies. Meta-analysis of these studies showed no significant difference between the drugs in preventing stroke (OR = 1.26 [0.73, 2.16]; p = 0.40, I2 = 0%; Supplementary Figure S18). The funnel plot revealed a symmetrical distribution, reflecting robust findings (Supplementary Figure S19). Subgroup analyses did not demonstrate any effect modification by the instance of drug administered (P-interaction: 0.73; Supplementary Figure S20) and by study design (P-interaction: 0.98; Supplementary Figure S21). Sensitivity analysis by removing Holm reduced the effect size (OR = 1.12 [0.62, 2.02]; p = 0.71, I2 = 0%; Supplementary Figure S22).
Myocardial infarction
Seven studies reported myocardial infarction. Meta-analysis of these studies showed no significant difference between the drugs in preventing myocardial infarction (OR = 1.13 [0.77, 1.66]; p = 0.53, I2 = 0%; Supplementary Figure S23). The funnel plot revealed a symmetrical distribution, reflecting robust findings (Supplementary Figure S24). Subgroup analysis did not demonstrate any effect modification by the instance of drug administered (P-interaction: 0.72; Supplementary Figure S25) or by study design (P-interaction: 0.55; Supplementary Figure S26). Sensitivity analysis did not reveal any significant differences.
Aspirin plus prasugrel versus aspirin plus clopidogrel
The PRISMA flowchart (Supplementary Figure S1) summarizes the search and selection process. Due to the limited data comparing aspirin plus prasugrel versus aspirin plus clopidogrel in patients undergoing CABG, we could identify and include only four relevant clinical trials in our analysis (1 RCT; 3 observational) [37, 38, 42, 43]. All the trials were at low risk of bias. A total of 817 patients were included in this comparison (n = 355 in the aspirin + prasugrel arm vs. n = 462 in the aspirin + clopidogrel arm). The mean follow-up time was 1 month. Patients had a mean age of 64.6 years, with 78.3% being males. The baseline characteristics of the patients included in this comparison have been outlined in Table II.
Table II
Summary of baseline characteristics in studies comparing prasugrel and clopidogrel effects in patients undergoing coronary artery bypass graft
[i] CABG – coronary artery bypass graft, Prasu – prasugrel, Clop – clopidogrel, BMI – body mass index, TRITON-TIMI – Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel–Thrombolysis in Myocardial Infarction, TIMI – thrombolysis in myocardial infarction, BARC – Bleeding Academic Research Consortium.
Platelet transfusion
Three studies reported on platelet transfusion. Meta-analysis of these studies showed that patients treated with prasugrel required significantly more platelet transfusions as compared to those treated with clopidogrel (OR = 1.88 [1.24, 2.87]; p < 0.01, I2 = 0%; Figure 2 A). The funnel plot presented a symmetrical distribution, indicating reliable results (Supplementary Figure S27).
Figure 2
The effect of prasugrel on (A) platelet transfusion and (B) all-cause mortality in CABG patients. Forest plots show the effect of prasugrel versus clopidogrel, in combination with aspirin on outcomes in patients undergoing coronary artery bypass graft surgery. Clopidogrel significantly required fewer platelet transfusions. No significant difference was observed for all-cause mortality
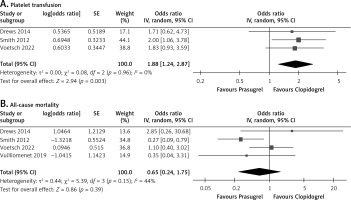
All-cause mortality
Four studies reported all-cause mortality. Meta-analysis of these studies showed no significant difference between the drugs in reducing all-cause mortality (OR = 0.65 [0.24, 1.75]; p = 0.39, I2 = 44%; Figure 2 B). The funnel plot revealed a symmetrical distribution, suggesting robust results (Supplementary Figure S28). Sensitivity analysis by removing Smith reduced heterogeneity and increased the effect size (OR = 1.05 [0.45, 2.48]; p = 0.91, I2 = 0%; Supplementary Figure S29).
Discussion
This meta-analysis offers a comprehensive evaluation of the comparative efficacy and safety of ticagrelor, prasugrel, and clopidogrel in the context of CABG. By computing data from 20 studies encompassing 9,585 patients, our analysis aims to address a critical aspect of cardiovascular care: the balance between reducing thrombotic events and minimizing bleeding complications, a key challenge in the management of antiplatelet therapy for patients undergoing CABG.
The existing literature on antiplatelet therapy predominantly addresses several critical areas. These include investigations into antiplatelet resistance [44], comparative studies assessing the efficacy of ticagrelor versus aspirin in the prevention of saphenous vein graft failure [45, 46], and evaluations of dual versus single antiplatelet therapy [47, 48]. Furthermore, research has focused on perioperative management strategies, particularly the optimal timing for discontinuation or continuation of aspirin [49]. To date, only one meta-analysis has compared the efficacy of aspirin plus ticagrelor or prasugrel versus aspirin plus clopidogrel [48]. Therefore, our study intends to fill these gaps by delivering a focused analysis of the efficacy of the three most common antiplatelet regimens administered in patients undergoing CABG.
Our study significantly advances the current literature by addressing key limitations of the previous meta-analysis, which combined ticagrelor and prasugrel despite their different mechanisms of action – ticagrelor being a reversible inhibitor and prasugrel an irreversible one [48]. By distinguishing between these agents and focusing on their specific pharmacological profiles, our analysis provides a more accurate and nuanced understanding of their respective roles in antiplatelet therapy. Furthermore, the earlier meta-analysis, which made a similar comparison, was limited by a small sample size (n = 2,429), restricting its statistical power and the robustness of its conclusions [48]. In contrast, with a sample size around four times that (n = 9,585), our study provides more reliable data, reducing statistical noise and strengthening the validity of our findings.
Unlike clopidogrel, which is a prodrug requiring metabolic activation and is subject to variability due to genetic polymorphisms in CYP2C19, ticagrelor directly and reversibly inhibits the P2Y12 receptor, providing consistent and potent platelet inhibition. The adoption of ticagrelor in the perioperative period has been historically tempered by concerns over its associated bleeding risk, particularly in surgical patients. Our meta-analysis provides robust evidence that this bleeding risk can be effectively managed by discontinuing ticagrelor at least 3 days before surgery [14]. This recommendation aligns with the pharmacokinetic properties of ticagrelor, which has a half-life of approximately 7 to 13 h, allowing for a significant attenuation of its antiplatelet effects within 3 days of discontinuation [50]. Notably, this finding challenges the traditional caution associated with ticagrelor in the surgical setting, suggesting that with appropriate perioperative management, ticagrelor can be both a safe and effective option for patients undergoing CABG.
Our analysis also explored the comparison between prasugrel and clopidogrel, albeit the data here was limited by a smaller sample size. Despite this limitation, the findings offer valuable insights. Prasugrel’s more potent antiplatelet effects provide significant thrombotic protection, particularly in patients with high-risk features such as diabetes or previous MI [51, 52]. This is in line with preclinical studies suggesting that prasugrel is approximately ten times more potent than clopidogrel in inhibiting platelet aggregation, preventing thrombus formation, and prolonging bleeding times [53]. However, this increased efficacy comes at the cost of a higher bleeding risk, as evidenced by the greater need for platelet transfusions in prasugrel-treated patients observed in our analysis. This trade-off highlights the critical need for individualized patient management, particularly in balancing the risk of bleeding against the benefits of potent platelet inhibition in the perioperative period.
The heterogeneity observed across trials in our meta-analysis, particularly regarding the timing of drug administration, follow-up duration, and patient characteristics, underscores the inherent complexity in managing antiplatelet therapy in CABG patients. The timing of drug discontinuation before surgery varied significantly among the studies, with some administering antiplatelet drugs before CABG and others after the procedure. This variation could influence the surgical bleeding risk and perioperative platelet function, leading to different outcomes. Moreover, follow-up durations varied widely among the trials, with studies administering DAPT after CABG generally having longer follow-ups, while those giving DAPT before CABG had shorter follow-ups, which aligns with the clinical focus of each. Short-term follow-up periods may capture immediate postoperative outcomes, such as bleeding and early graft occlusion, but may miss long-term benefits or complications, such as late graft patency, MI, and overall survival. This variability highlights the need for more standardized approaches in future studies to allow for better comparability of results.
One of the strengths of our analysis is the prioritization of the BARC (Bleeding Academic Research Consortium) criteria for assessing bleeding outcomes, whenever possible. The BARC criteria are considered more sensitive in detecting bleeding events compared to other methods like TIMI or PLATO, which were used in some of the included studies [17, 24, 31, 34]. By standardizing bleeding assessments, we minimized variability and enhanced the reliability of our findings. This approach provides a clearer understanding of the comparative safety and efficacy of these antiplatelet agents, particularly in the context of CABG, where bleeding complications are a major concern.
Despite these strengths, several limitations of this meta-analysis must be acknowledged. Sixteen of the nineteen included studies were observational, which introduces potential selection biases and confounding. While we employed rigorous standard methods to assess study quality and minimize bias, the inherent limitations of observational studies cannot be fully eliminated. Furthermore, the mean duration of follow-up was only 1 year, limiting the ability to fully assess the long-term outcomes in CABG patients. Future studies with longer follow-up durations are needed to address this limitation. Additionally, the relatively small sample sizes in studies comparing prasugrel to clopidogrel limit the generalizability of our findings concerning prasugrel’s safety and efficacy in the CABG setting. Moreover, variability in bleeding assessment criteria across studies may have influenced our pooled estimates of bleeding risk, despite our efforts to standardize outcomes using the BARC criteria. Lastly, our inability to analyze the number of days before drug discontinuation as a continuous variable, due to the categorical nature of the data provided by the studies, further limited our analysis. This limitation underscores the need for future studies to report data in a more granular and continuous manner, which would allow for more precise and informative analyses.
In the broader landscape of current research, our findings contribute significantly to the ongoing debate regarding the optimal antiplatelet strategy for patients undergoing CABG. Recent guidelines from the American College of Cardiology/American Heart Association (ACC/AHA) recommend DAPT in certain high-risk patients, but the choice of P2Y12 inhibitor and the timing of discontinuation before surgery remain areas of active investigation [54]. Our meta-analysis provides evidence that supports a more nuanced approach to the use of ticagrelor and prasugrel perioperatively, potentially informing future updates to these guidelines.
Future research should focus on large-scale trials that investigate long-term outcomes such as graft patency and recurrent cardiovascular events. Additionally, exploring the role of genetic testing to personalize antiplatelet therapy, particularly in clopidogrel-treated patients with CYP2C19 polymorphisms, may help optimize treatment strategies [55]. Additionally, long-term studies focusing on patient survival, graft patency, and recurrent cardiovascular events are essential for a comprehensive understanding of the long-term implications of different antiplatelet strategies.
Conclusions
This meta-analysis provides strong evidence that post-CABG ticagrelor administration with aspirin is associated with reduced all-cause mortality in patients compared to clopidogrel with aspirin. Importantly, the bleeding risk associated with ticagrelor with aspirin can be effectively managed by discontinuing the antiplatelet therapy at least three days before surgery. Additionally, ticagrelor demonstrated marginally better outcomes in preventing re-bleeding and requiring fewer platelet transfusions, highlighting its clinical benefits in these areas. Although the included studies are predominantly observational, which limits causal inferences, the large sample size contributes to reduce some degree of residual confounding. The associated bleeding risk can be effectively managed through careful perioperative planning. Prasugrel, while offering potent antiplatelet effects, presents a greater bleeding risk, necessitating cautious use and a tailored approach to drug discontinuation before surgery. These findings underscore the importance of individualized antiplatelet therapy in CABG patients and provide a foundation for refining clinical guidelines to optimize patient outcomes in this high-risk population. Future research should focus on large, multi-center trials with standardized methodologies to validate these findings and explore the potential role of genetic testing and other biomarkers in personalizing antiplatelet therapy for CABG patients.



