Introduction
There is growing interest in lipid-lowering dietary supplements with ancillary protective effects on vascular health. The cost-benefit ratio of using lipid-lowering drugs in subjects with moderate hypercholesterolaemia and low added cardiovascular risk is questionable [1]. Consequently, the development of clinically tested lipid-lowering nutraceuticals could have a significant preventive impact on the general population.
Among the most clinically studied lipid-lowering nutraceuticals, red yeast rice [2] and omega-3 polyunsaturated fatty acids (PUFAs) [3] stand out.
Red yeast rice is an extensively tested dietary supplement capable of reducing low-density lipoprotein (LDL)-cholesterol by reversibly inhibiting 3-hydroxy-3-methylglutaryl coenzyme A reductase, the key enzyme in cholesterol biosynthesis in the liver [4]. In 2011, the European Food Safety Agency (EFSA) approved a health claim related to the ability of red yeast rice to improve LDL-cholesterol, linked to the content of 10 mg monacolin K per daily dose of the dietary supplement [5]. Although a large meta-analysis of randomized clinical trials supported the safety of red yeast rice-containing supplements [6] in 2018, the EFSA suggested that its absolute safety could not be confirmed [7]. As a result, in 2022, the European Commission decided that red yeast rice-containing supplements in the EU should contain less than 3.0 mg total monacolins per daily dose [8]. Whether this dose is related to a significant improvement in LDL-cholesterol and other cardiovascular risk parameters remains to be determined.
Eicosapentaenoic (EPA) and docosahexaenoic (DHA) omega-3 fatty acids are essential fatty acids whose dietary intake is inversely associated with the risk of all-cause mortality in the general population [9] a meta-analysis of 11 prospective studies involving 371,965 participants and 31,185 death events showed that all-cause mortality for high versus low omega-3 PUFAs intake was 0.91 (95% CI: 0.84–0.98), and for EPA and DHA intake it was 0.83 (95% CI: 0.75–0.92) and 0.81 (95% CI: 0.74–0.95), respectively. In the dose-response analysis, each 0.3 g/day increment in omega-3 PUFAs was associated with 6% lower risk of all-cause mortality (RR = 0.94, 95% CI: 0.89–0.99). Thus, the EFSA suggests that 250 mg of EPA-DHA is needed to maintain adequate cardiovascular health [10]. Even if high dosages of EPA and DHA are also associated with significant reduction in plasma triglycerides (TG), blood pressure and high-sensitivity C-reactive protein (hsCRP) [11], the effect of small supplemented doses should also be confirmed.
The aim of our study was to test the short-term effect of a combined nutraceutical including red yeast rice and fish oil standardized in PUFAs on plasma lipids, hsCRP, and endothelial function in healthy subjects.
Material and methods
This was a randomized, double-blind, placebo-controlled, parallel-group clinical study that enrolled a sample of 60 Caucasian healthy subjects with suboptimal LDL-cholesterolaemia recruited from March to September 2018 in the outpatient Lipid Clinic of the S. Orsola Malpighi University Hospital, Bologna, Italy.
Inclusion criteria of the trial were as follows: age ranging between 30 and 75 years, with suboptimal LDL-C plasma level (LDL-C > 115 mg/dl and < 190 mg/dl), and an estimated 10-year cardiovascular risk < 5% according to the SCORE risk charts [12]. Exclusion criteria included previous history of CV disease, obesity (body mass index (BMI) > 30 kg/m2), TG > 400 mg/dl, diabetes mellitus, uncontrolled hypertension (i.e. systolic and diastolic blood pressures > 190/100 mm Hg), uncontrolled hypothyroidism, known myopathies, kidney or liver diseases, the use of any lipid-lowering medication or nutritional supplement, known intolerance to red yeast rice or PUFAs, alcoholism, pregnancy and breastfeeding, and any medical or surgical condition causing complex or inconstant adhesion to the study protocol.
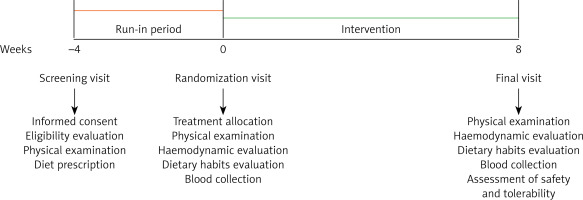
The enrolled subjects adhered to a standard Mediterranean diet for 4 weeks before randomization. The intervention period lasted 8 weeks. At each follow-up visit, subjects were evaluated for clinical status and by the execution of a physical examination and laboratory and haemodynamic analyses. The study timeline is described in detail in Figure 1.
The study fully complied with the ethical guidelines of the Declaration of Helsinki and with the International Council for Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) Harmonized Tripartite Guideline for Good Clinical Practice (GCP). The study protocol was approved by the Ethical Committee of the University of Bologna. All subjects signed a written informed consent form to participate.
Treatment
After a 1-month period of diet standardization, the enrolled subjects were randomized to receive either an indistinguishable softgel of placebo or the investigational product containing omega-3 PUFAs, red yeast rice, astaxanthin, folic acid, coenzyme Q10, and vitamin B6 (Table I).
Table I
Quantitative composition of the active treatment and the related placebo tested in the clinical study
The study products were released and supplied by Meda Pharma S.p.A. (Monza, MB, Italy), a site compliant with EN UNI ISO 9001:2015 and the requirements of HACCP (Hazard Analysis and Critical Control Points).
At the time of randomization, subjects were provided with boxes containing 60 softgels each.
Randomization was performed centrally using computer-generated codes. Participants and investigators were blinded to the group assignment. The randomization codes were kept in a sealed envelope that was opened after study completion and data analysis.
For the entire duration of the study, the subjects were instructed to take a softgel of the assigned treatment once daily, at about the same time each day, preferably in the evening.
At the end of the clinical trial, all unused pills were retrieved for inventory. Participants’ compliance was assessed by counting the number of returned pills.
Assessments
Clinical data and anthropometric measurements
Information gathered in the subjects’ history included the presence of cardiovascular and other systemic diseases, allergies, and medications. Validated semi-quantitative questionnaires including the Food Frequency Questionnaire (FFQ) were used to assess demographic variables, recreational physical activity, and dietary and smoking habits [13, 14].
Laboratory analyses
Biochemical analyses were conducted on venous blood collected after an overnight fast of at least 12 h. Serum was obtained by adding disodium ethylenediaminetetraacetate (Na2EDTA) (1 mg/ml) and centrifuging the blood at 3000 rpm for 15 min at 25°C. Following centrifugation, laboratory analyses were promptly carried out by trained personnel using standardized methods [15, 16]. The following parameters were directly assessed: total cholesterol (TC), triglycerides (TG), high-density lipoprotein cholesterol (HDL-C), apolipoprotein B-100 (apoB-100), apolipoprotein AI (apo-AI), fasting plasma glucose (FPG), creatinine, γ-glutamyl transferase (GGT), alanine (ALT), aspartate transaminase (AST), high-sensitivity C-reactive protein (hs-CRP), and creatine phosphokinase (CPK).
LDL-C was obtained by the Friedewald formula. Non-HDL cholesterol (non-HDL-C) resulted from the difference between TC and HDL-C. The glomerular filtration rate (eGFR) was estimated by the Chronic Kidney Disease Epidemiology Collaboration (CKD-epi) equation [17].
Blood pressure measurements
Blood pressure (BP) was measured in accordance with the recommendations of the International Guidelines for the Management of Arterial Hypertension [18]. Resting systolic (SBP) and diastolic BP (DBP) were measured with a validated oscillometric device and a cuff of the appropriate size applied to the right upper arm. To improve detection accuracy, 3 BP readings were sequentially obtained at one-minute intervals. The first reading was discarded, and the average between the second and the third reading was recorded as a study variable.
Endothelial reactivity
Endothelial function of the arterial vasculature is an important early marker of atherosclerosis, reflecting the ability of the endothelial layer to release nitric oxide (NO), modulating smooth muscle tone in the arterial wall of the conduit arteries [19].
Following the current guidelines [20], during the clinical study endothelial function was evaluated using Endocheck® (BC Biomedical Laboratories Ltd, Vancouver, BC, Canada), a method embedded within the Vicorder® device, which guarantees very good intra- and inter-operator reliability [21]. The measurement was carried out with subjects in a supine position and in abstinence from cigarette smoking and caffeinated beverages for at least 12 h. After a 10-minute rest, the brachial pulse volume (PV) waveforms were recorded at baseline for 10 s and during reactive hyperaemia. The BP cuff was inflated to 200 mm Hg for 5 min, and PV waveforms were recorded for 3 min after the cuff was released. PV displacement was calculated as a percentage change in the PV waveform area, comparing waveforms before and during hyperaemia using the equation PV2/PV1, where PV1 represents PV at the baseline and PV2 represents PV during hyperaemia [22]. This test has already been largely used to detect the effect of dietary supplements on endothelial function [23, 24].
Assessment of safety and tolerability
The study assessed the safety and tolerability by continuously monitoring adverse events, clinical safety, laboratory findings, vital sign measurements, and physical examinations. The principal investigator appointed a blinded, independent expert clinical event committee to categorize any adverse events that might occur during the trial as not related, unlikely to be related, possibly related, probably related, or definitely related to the tested treatment [25].
Statistical analysis
The sample size was calculated with a focus on the change in LDL-C. To detect the mean change in LDL-C at 8 weeks of 12 mg/dl with a power of 0.90 and an α error of 0.05, a total of 28 subjects per group were required. Including a 5% dropout rate, the study involved a total sample size of 60 subjects (30 subjects per arm).
The normality distribution of the studied variables was assessed using the Kolmogorov-Smirnov test. Non-normally distributed variables underwent log transformation before further statistical testing. The baseline characteristics of the population were compared using an independent Student’s t-test and χ2 test followed by Fisher’s exact test. Between-group differences were evaluated through repeated-measures ANOVA followed by Tukey’s post hoc test. All data are represented by means and related standard deviations. The data were analysed utilizing the intention-to-treat approach through the Statistical Package for Social Sciences (SPSS) 22.0, Windows version. All tests were 2-sided, and a p level of < 0.05 was considered significant for all analyses.
Results
Sixty subjects (32 women, 28 men) were enrolled. The main characteristics of the enrolled subjects at the screening visit are shown in Table II.
Table II
Main characteristics of the enrolled subjects at the screening visit
No ADR or drop-out was registered during the study. Four subjects in the active treatment group reported a mild fishy aftertaste after product assumption. Mean compliance was 95% in both groups.
After the run-in period, no statistically significant differences were detected among the groups that were well matched for all the considered variables at baseline (Table III). No statistically significant changes were recorded in the dietary habits (i.e. in total energy and macronutrient intake) of the enrolled individuals from randomization until the end of the study. Similarly, self-reported physical activity did not change for the participants over the course of the clinical trial.
Table III
Effect of the tested product and placebo on anthropometric and biochemistry parameters
Waist circumference, LDL, and total cholesterol and apo levels significantly decreased both in the placebo and combined nutraceutical groups versus baseline. However, LDL and total cholesterol as well as the apoB and hsCRP plasma levels significantly improved versus placebo in the tested nutraceutical only (Table III).
None of the tested products was associated with a significant change versus baseline blood pressure values. Pulse change was significantly increased by the experimental product, both versus baseline and placebo (Table IV).
Table IV
Effect of the tested product and placebo on haemodynamic parameters
Discussion
In our randomized, double-blind, placebo-controlled, parallel-group clinical study carried out on 60 Caucasian healthy subjects with suboptimal LDL-cholesterolaemia we observed that combined dietary supplements containing red yeast rice (2.8 mg monacolins) and PUFAs (588 mg of fish oil, standardized in PUFAs: 350 EPA, 45 mg DHA) had a positive impact on plasma lipid pattern versus baseline and versus placebo. In particular, after 8 weeks of supplementation we observed a 17.3 ±3.4% reduction of LDL-C, a 12.1 ±2.2% reduction of TC, a 22.3 ±4.3% reduction of apoB, a 14.9 ±1.8% reduction of hsCRP, and a significant improvement of pulse volume change by 5.0 ±0.9%
In a previous observation, we evaluated the effects of a combined nutraceutical with red yeast rice (5 mg monacolin K) and PUFAs (610 mg of fish oil, standardized in PUFAs: 183 mg EPA, 122 mg DHA) in a multi-centre clinical trial involving 107 subjects with suboptimal LDL-C levels and in the context of metabolic syndrome. After 8 weeks of treatment, the results showed a significant decrease in LDL-C (–37.5 mg/dl; –22 ±3%), TG (–19.5 mg/dl; –9 ±5%), and non-HDL-C (–21 ±3%) and a significant increase in HDL-C (+1.5 ±0.5%) (p < 0.001 for all), without changes in safety parameters. In particular, the study highlighted a greater decrease in TG levels only in subjects with baseline TG > 500 mg/dl, who reached 11% reduction (p < 0.001 vs. subjects with baseline TG < 150 mg/dl) [26].
Other colleagues, testing a similar formulation including red yeast rice and PUFAs (525 mg of fish oil, standardized in PUFAs: 294 mg EPA, 147 mg), observed an LDL-C and TC reduction of 17.3% and 12.0%, respectively [27].
Considering that in both trials the tested dose of monacolin K was about double that of the one currently tested in our study, the overall effect in term of blood lipids reduction was similar in all studies.
On the other hand, we find of interest the impact of the tested formulation on hsCRP, a well-known inflammatory biomarker strictly related to cardiovascular risk [28], as well as on pulse volume change, where even a small improvement in endothelial function is associated with a significant improvement in cardiovascular disease risk. In particular, a 1% improvement in endothelial function has been estimated to be associated with a 12% reduction in cardiovascular disease events [29]. The anti-inflammatory effect and the positive effect of both red yeast rice [30] and omega-3 PUFAs [31] are well-known but both for higher doses than the one tested in this trial. Consequently, we should argue that the combined effect of both components in a single softgel could have improved their individual efficacy.
While more extended investigations are necessary to validate the observed outcomes, the duration of this study suffices to identify the lipid-lowering effectiveness of the tested product. It is essential to acknowledge the limitations in our study, including the brief duration and the relatively small sample size, even though the study was adequately powered for the primary outcome measure. It is crucial to note that our findings may not be universally applicable to subjects with obesity because individuals with a BMI ≥ 30 kg/m2 were excluded from the study. Additionally, due to the study’s relatively short duration, we cannot ascertain whether the observed efficacy and safety data would hold true in the long term [32]. Of course, the other minor components of the tested combined nutraceutical could have also contributed to its global effect, but the design of the study cannot derive any conclusion about that.
In conclusion, the tested combined dietary supplement containing red yeast rice and fish oil containing PUFAs was very well tolerated and significantly improved LDL-C, TC, apoB, hsCRP, and endothelial function in healthy subjects with suboptimal LDL-cholesterolaemia.