Introduction
Acute myocardial infarction (AMI) is a common manifestation of cardiovascular heart disease and a leading contributor of morbidity and mortality in the United States [1]. It is defined by the presence of acute myocardial injury, detected by a rise and/or fall in serum cardiac biomarkers (high-sensitivity troponin) in conjunction with evidence of myocardial ischemia on electrocardiogram (ECG), imaging, or angiography [2]. In addition, AMI is further clinically classified by the presence or absence of ST-segment elevation on ECG and this has implications for patient triage and acute management. While the diagnosis and treatment of an ST-segment elevation myocardial infarction (STEMI) are unequivocal (i.e. emergent coronary angiography), in the case of a non-ST-segment elevation myocardial infarction (NSTEMI) they remain a clinical challenge. Further, whereas STEMI and type-1 NSTEMI (T1AMI) are precipitated by coronary atherosclerotic plaque disruption and atherothrombosis, type-2 NSTEMI (T2AMI) is not caused by atherosclerotic CAD and may be due to myocardial ischemia from a plethora of etiologies such as coronary spasm or microvascular dysfunction, non-atherosclerotic coronary dissection, or oxygen supply/demand imbalance due to another acute medical cause [2, 3]. Common triggers are sepsis, anemia and tachyarrhythmia [4, 5].
High-sensitivity troponin is an ultra-sensitive marker for myocardial injury, which has revolutionized the recognition of AMI by providing earlier detection capabilities and improving the accuracy of the test, in part by improving upon the negative predictive value [6, 7]. This diagnostic tool has led to an increase in the incidence of NSTEMI relative to STEMI [8, 9]. However, neither a clear consensus in the exact definition of T2AMI nor formal guidelines for its diagnosis exist, which may lead to clinician misclassification of this entity as T1AMI [3, 10]. Consequently, this causes difficulties in triage and further management of these two clinically separate entities. T1AMI patients generally benefit from an urgent left-heart catheterization for treatment of their atherothrombotic lesion. In contrast, T2AMI patients are treated for their underlying medical illness that is presumably causing supply/demand ischemia and the role of coronary angiography is unknown and may at times be inappropriate [11]. This divergence in management of NSTEMI patients underscores the need for accurate and prompt diagnosis and triage upon presentation.
T2AMI patients tend to be older, more often women, and more likely to have a history of multiple co-morbidities [12]. Clinical laboratory variables, such as the ratio of low-density lipoprotein to high-density lipoprotein (L/H), N-terminal pro-B-type natriuretic peptide/troponin I (P/T), and presence of regional wall motion abnormalities (RWMA) on cardiac echocardiogram may also be useful for distinguishing T1AMI from T2AMI [13–16]. Whether any of these clinical variables independently predict the likelihood of T1AMI from T2AMI is unknown. The purpose of this study was to investigate the predictive effect of demographic and clinical variables on predicting NSTEMI subtypes in patients presenting to the ED with ischemic symptoms.
Material and methods
We performed a retrospective study of patients presenting to the ED in an urban, university affiliated community hospital, which satisfied the following parameters: 1) acute ischemic symptoms (shortness of breath and chest pain) suspicious for AMI, 2) diagnosed with an NSTEMI (according to the Fourth Universal Definition of Myocardial Infarction), and 3) underwent coronary angiography between October 1, 2016 and June 31, 2018 [2]. We excluded patients with high-risk features (dynamic ST-segment elevations, cardiogenic shock, refractory chest pain, and unstable ventricular arrhythmias).
A diagnosis of T1AMI was given if coronary angiography displayed the presence of a culprit atherothrombotic lesion with at least one critical stenosis for which a coronary intervention was performed. A diagnosis of T2AMI was given if results of angiography demonstrated non-obstructive coronary arteries. Troponin I (ng/ml) obtained through a contemporary assay was abstracted from medical records. The L/H and P/T ratios were calculated by taking the ratio of low-density lipoprotein (LDL) to high-density lipoprotein (HDL) and proBNP to troponin I, respectively. Transthoracic echocardiogram was performed during the index hospitalization prior to coronary angiography utilizing a protocol in line with the American Society of Echocardiography/Intersocietal Accreditation Commission and it was interpreted by a board-certified cardiologist. Left ventricular ejection fraction (LVEF) was measured utilizing the 2D, bi-plane or single plane method of disks at the discretion of the interpreting cardiologist. Regional wall motion abnormalities were assessed based on the 17-segment model and classified as positive if hypokinesis, akinesis, or dyskinesis was present [17].
The study received expedited approval by the institutional review board at New York Presbyterian-Brooklyn Methodist Hospital in September 2018.
Statistical analysis
Continuous and categorical variables are presented as mean with one standard deviation or median with interquartile range, as appropriate, and were compared with Student’s t-test for means, the Mann-Whitney test for medians, and the two-sided z-test or χ2 test for proportions. Univariate analysis of various clinical variables of interest were compared with odds ratios. Binary logistic regression was used to determine the independent predictors of a T1AMI. The following demographic and clinical covariates were simultaneously included in the model: age, sex, race, body mass index (BMI), hypertension, diabetes, end-stage renal disease (ESRD), heart failure, family history of CAD (FH of CAD), prior myocardial infarction (MI), LVEF, L/H ratio, presence of RWMA, and percentage change of troponin I. A receiver operating characteristics (ROC) curve was generated based on the probability table from the logistic regression model. The significance level was set at p < 0.05 (two-sided). Tables displaying the odds ratios from univariate analysis were generated with Review Manager (RevMan) computer program (Version 5.4. The Cochrane Collaboration, 2020). All analyses were performed with IBM SPSS Statistics for Mac, version 24 (IBM Corp., Armonk, N.Y., USA).
Results
Between October 1, 2016 and June 31, 2018, 1,123 patients were identified in the institutional registry who presented to the ED with ischemic symptoms suggestive for NSTEMI. A hundred and thirteen patients were duplicate entries. In consideration for the assessment of RWMA, 464 patients were excluded because an echocardiogram before left-heart catheterization or either imaging modality was not performed. A total of 546 patients were enrolled in the study. Through coronary angiography, 426 patients were found on coronary angiography to have T1AMI, whereas 120 patients had T2AMI. Baseline characteristics, by type of NSTEMI, are displayed in Table I. In summary, patients with T1AMI were more likely to present with chest pain, be male, white, have diabetes mellitus, a positive family history of CAD and prior MI, a higher LDL/HDL ratio, higher levels of serum troponin, and more likely to have RWMA on echocardiography.
Table I
Comparative table of baseline demographics by type of non-ST segment myocardial infarction
[i] BMI – body mass index, CABG – coronary artery bypass graft, FamHx of CAD – family history of coronary artery disease, HDL – high-density lipoprotein, LVEF – left ventricular ejection fraction, LDL – low-density lipoprotein, MI – myocardial infarction, pro-BNP – pro-basic natriuretic protein, RWMA – regional wall motion abnormality.
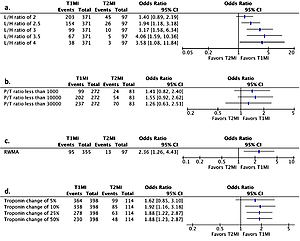
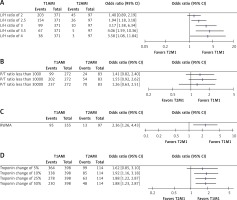
L/H ratio > 2.5 (OR = 1.94 (1.18, 3.18), p = 0.008), troponin change > 25% (OR = 1.88, (1.22, 2.87), p = 0.002), and presence of RWMA (OR = 2.36 (1.26, 4.43), p = 0.006) were statistically significantly associated with the T1AMI group, while the P/T ratio was not (Figure 1).
Figure 1
Univariate analysis of various clinical variables predictive of type-1 myocardial infarction. A – LDL/HDL (L/H) ratio. B – proBNP/troponin I (P/T) ratio. C – Regional wall motion abnormality (RWMA). D – Percentage change in troponin I
In binary logistic regression analysis, age (OR per year = 1.03 (1.00, 1.05), p = 0.03), prior MI (OR = 3.50 (1.68, 7.22), p = 0.001), L/H > 2.0 (OR = 1.55 (1.12, 2.13), p = 0.007), percentage change in troponin I > 25% (OR = 2.54 (1.38, 4.69), p = 0.003), and RWMA (OR = 3.53 (1.46, 8.54), p = 0.004) were independent predictors of T1AMI, while sex, race, BMI, hypertension, ESRD, heart failure, FH of CAD, HbA1c, and LVEF were not (Table II).
Table II
Independent predictors of type-1 NSTEMI
[i] BMI – body mass index, FH of CAD – family history of coronary artery disease, ESRD – end-stage renal disease, HbA1c – hemoglobin A1c, L/H – low-density lipoprotein to high-density lipoprotein ratio, LVEF – left ventricular ejection fraction, MI – myocardial infarction, RWMA – regional wall motion abnormality.
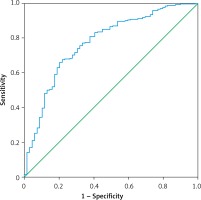
The overall accuracy of the prediction algorithm was 81% (p < 0.001) (Table III). The area under the ROC curve was 0.77 (Figure 2).
Discussion
The major findings from this single-institution, retrospective study of angiographic data obtained from left-heart catheterization from 549 patients with an index NSTEMI are as follows: 1) Patients with T1AMI were more likely to be male, white, have diabetes mellitus, a positive family history of CAD and prior MI, a higher LDL/HDL ratio, higher levels of serum troponin, present with chest pain, and more likely to have RWMA on echocardiography. 2) Clinical variables such as age, prior MI, L/H ratio > 2.0, percentage change in troponin I > 25%, and presence of RWMA on echocardiogram were significant predictors of T1AMI in patients presenting with ischemic symptoms to the ED. 3) The prediction algorithm had an accuracy of 81% and an area under the curve of 77%.
Prior studies have reported that T2AMI patients tend to be older, more often women, and more likely to have a history of multiple co-morbidities [12, 18]. Although our cohort of T2AMI patients also tended to be more often women with lower triglyceride levels and lower levels of serum troponin, our cohort exhibited decreased prevalence of cardiac co-morbidities. This discrepancy in prevalence of cardiac risk factors may be due to our T2AMI cohort being younger compared to other studies since a similarly aged cohort also did not detect differences in prevalence of cardiac co-morbidities [19]. Misdiagnosis of T2AMI is also commonplace and may be another reason for the discrepancies encountered in the literature. To eliminate the potential for misclassification from subjective clinical data that may be equivocal, our study was limited to angiography-proven T2AMI patients [16, 20].
Although the role of peak troponin I and hyperlipidemia in predicting the likelihood of a T1AMI has been previously reported, our finding that age, history of prior MI, L/H ratio > 2.0, percentage change in troponin I > 25%, and presence of RWMA on echocardiogram are predictive of a T1AMI is novel as these variables have not been previously demonstrated to be independent discriminators of a T1AMI from a T2AMI diagnosis [19].
Age was a significant predictor of a T1AMI rather than a T2AMI in our cohort, which is contrary to prior reports [18]. This discrepancy is likely due to the inclusion criteria of our study, which focused on patients presenting with ischemic symptoms such as chest pain and shortness of breath. It is well documented that the incidence of sepsis, anemia, arrhythmia, heart failure, stroke, tachyarrhythmia and other etiologies of supply/demand ischemia increases with age and we acknowledge that age may not be validated as a predictor of T2AMI in those presenting without symptoms of ischemia [4, 5].
Prior MI and change in troponin I were some of the most robust predictors for distinguishing a T1AMI from T2AMI. In addition, the ratio of low-density lipoprotein to high-density lipoprotein (L/H) has been shown to be a predictor of cardiovascular events in coronary artery disease and a proxy of the vulnerability of plaque rupture [13], [14]. It is therefore a sensible surrogate marker for acute plaque rupture and T1AMI.
Importantly, although sex was a significant variable on univariate analysis, it was not shown to be an independent predictive of a T2AMI. A prior report has documented the utility of the P/T ratio in discriminating coronary vs. non-coronary causes of troponin elevation in the ED setting. Our study was not able to verify the benefit of this ratio in confirming T1AMI vs. T2AMI. Our study included a broader cohort of patients including those with congestive heart failure and CABG, whereas the prior report did not, which may account for the discrepancy in the findings [21].
A clear consensus in the exact definition of T2MI or formal guidelines in its diagnosis are lacking. Presently, misclassification of T2AMI as either T1AMI or a missed diagnosis of T2AMI may be as high as 29.7% and 63%, respectively [20, 22]. The role of coronary angiography in this cohort may be unclear and at times inappropriate. T2AMI has been associated with worse outcomes, and it remains to be seen if correction of the misclassification and greater focus on the treatment of the underlying etiology of the supply/demand mismatch will improve outcomes in this cohort [23]. The above-mentioned variables may be helpful clues to the assessing physician to assist in the correct classification of T1AMI and T2AMI in those presenting with ischemic symptoms. The broader applicability to those without ischemic symptoms requires further investigation.
We recognize important limitations to our study design, observations and conclusions. First, the relatively small cohort of patients included in this single-center study may limit the generalizability to the US population as a whole. Second, data were collected retrospectively from electronic medical records and were not recorded in a standardized and systematic manner, thus obscuring confounders that cannot be optimally controlled for. Third, the applicability of our findings to a broader T2AMI cohort is unclear as our study considered only patients presenting to the ED with ischemic symptoms and selected for coronary angiography. A prospective evaluation of all patients presenting with NSTEMI is necessary to validate our findings.
In conclusion, in the present single-institution, retrospective study of angiographic data obtained from left-heart catheterization from patients with an index NSTEMI (after adjustment for important baseline demographic and clinical variables), age, prior MI, L/H ratio, percentage change in troponin I, and presence of RWMA on echocardiogram were significant predictors of T1AMI in patients presenting with ischemic symptoms to the ED. Further studies are needed to validate this prediction algorithm in larger and broader cohorts.