Hematocrit increase with sodium-glucose co-transporter-2 (SGLT-2) inhibitors has been proposed as a potential mechanism implicated in the well-established cardio- and reno-protection with this drug class [1]. According to a recent, large meta-analysis of randomized controlled trials in a total of 14,478 participants, SGLT-2 inhibitors led to a significant increase in hematocrit by 1.32% and in hemoglobin by 0.56 g/dl [2]. Increases in erythropoiesis and red-blood cell count, along with decreases in hepcidin, ferritin and transferrin saturation levels, have been observed [3–5]. In the present sub-analysis of a real-world study performed in the context of the COVID-19 pandemic we sought to determine the effect of different SGLT-2 inhibitors on hematocrit levels and their association with established cardiovascular risk factors.
This is a sub-analysis of a single-center, prospective, observational study, conducted in the Diabetes Center of the Second Propedeutic Department of Internal Medicine, Aristotle University of Thessaloniki, General Hospital “Hippokration”, between January 2020 and August 2021. The study protocol was approved by the Ethics Committee of the Medical School, Aristotle University of Thessaloniki (protocol number: 4/17.7.2019). Study procedures were performed strictly in accordance with the principles outlined in the Declaration of Helsinki. The study has been registered in the International Standard Registered Clinical/soCial sTudy Number (ISRCTN) registry (registration number ISRCTN88851713). Our initial study aims to identify the effect of long-term treatment with SGLT-2 inhibitors on ambulatory arterial stiffness indices.
Adult subjects aged 18–75 years, with an established diagnosis of type 2 diabetes mellitus (T2DM) (≥ 12 months), glycated hemoglobin (HbA1c) value ranging between 6.55 and 10.0%, stable antidiabetic and antihypertensive treatment over the last 6 months and an indication for the initiation or addition of new antidiabetic therapy with a SGLT-2 inhibitor according to the treating physician’s discretion were eligible to participate, after providing written informed consent. Blood sampling was performed for all patients, after overnight fasting, both at visit 1 (prior to initiation of a SGLT-2 inhibitor) and at visit 2 (planned to be performed 6 months after visit 1).
Enrolled participants were prescribed either dapagliflozin 10 mg or empagliflozin 10 mg once daily, according to the treating physician’s clinical discretion. The follow-up visit was planned to be performed 6 months after initiation of an SGLT-2 inhibitor.
Continuous variables are presented as mean ± standard deviation (SD) or median ± interquartile range (IQR), according to normality of distribution, while categorical variables are presented as relative frequencies and percentages (n, %). The Shapiro-Wilk test was used to test for normality, and the F-test was used to test for equal variances between the two groups. In the case of normal distributions, we performed hypothesis testing using the one-tailed paired t-test, while in the opposite case we used the one-tailed Wilcoxon signed rank test. The Pearson correlation coefficient was used to assess the correlation of variables of interest. A p-value < 0.05 was considered statistically significant. R software was used for all statistical analyses.
A total of 51 subjects participated in the study. Because the study was conducted during the COVID-19 pandemic, finally, a total of 46 subjects with T2DM were included, since 3 discontinued treatment and 2 were lost to follow-up.
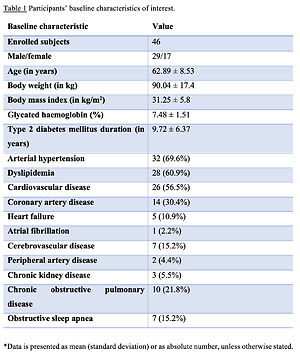
Age of enrolled subjects was 62.89 ±8.53 years, body weight was 90.04 ±17.4 kg, body mass index was 31.25 ±5.8 kg/m2, while T2DM duration was 9.72 ±6.37 years. Thirty patients were prescribed dapagliflozin and 16 were initiated on empagliflozin. Participants’ baseline characteristics are summarized in Table I. Despite the prespecified date of the follow-up visit, COVID-19 pandemic regulations imposed by the National Health Authorities led to a significant delay in follow-up visits. Therefore, the mean treatment duration and follow-up period was 9.98 ±3.27 months.
Table I
Participants’ baseline characteristics of interest
A significant proportion of participants had concomitant hypertension (69.5%) and dyslipidemia (60.8%), while 56.5% had pre-existing cardiovascular disease (Table I). Regarding their antidiabetic treatment prior to enrollment, 93.5% of patients received metformin, 28.3% received insulin, 32.6% were prescribed glucagon-like peptide-1 (GLP-1) receptor agonists and 39.1% were administered dipeptidyl-peptidase-4 (DPP-4) inhibitors. Detailed information regarding patients’ pharmacotherapy prior to inclusion in the study is provided in Table II.
Table II
Participants’ drug treatment prior to enrollment in the study
Hematocrit levels increased from 41.3 ±3.7 to 43.6 ±3.9% (p < 0.001), while hemoglobin increased from 13.6 ±1.3 to 14.4 ±1.3 g/dl (p < 0.001) with SGLT-2 inhibitor treatment. Empagliflozin led to a significant increase in hematocrit by 1.58% (p = 0.011) and in hemoglobin by 0.49 g/dl (p = 0.014), while dapagliflozin induced a significant increase in hematocrit by 2.7% (p < 0.001) and in hemoglobin by 0.89 g/dl (p < 0.001). History of background cardiovascular disease did not significantly affect the observed effect since hematocrit increased significantly both in patients with (p < 0.001) and without (p = 0.001) baseline cardiovascular disease.
Of note, when we addressed the potential correlation of hematocrit (and hemoglobin) with other established cardiovascular risk factors, we did not find a significant correlation with body weight (p = 0.57), office systolic and diastolic blood pressure (p = 0.89 and 0.18, respectively), estimated glomerular filtration rate (p = 0.76), uric acid (p = 0.74), proteinuria (p = 0.2), glycated hemoglobin (p = 0.79), total cholesterol (p = 0.81), low-density lipoprotein cholesterol (p = 0.36) or triglycerides (p = 0.74). In addition, no significant correlation with ambulatory arterial stiffness indices (such as pulse wave velocity, augmentation index and central pulse pressure) or with 24-hour ambulatory blood pressure was found. Interestingly, a marginally non-significant positive correlation with high-density lipoprotein cholesterol was found (r = 0.27, p = 0.06). Finally, no significant correlation was detected between the change in hematocrit or hemoglobin and any of the drug classes utilized prior to participation in the study. This is of great importance, since drug classes such as diuretics might produce an increase in hematocrit and hemoglobin due to plasma volume contraction.
The main limitation of the present study is the small sample size. The study design (observational study) represents an additional limitation. Finally, our study was conducted in the context of the COVID-19 pandemic, which has a major impact on patients’ quality of life, due to social distancing, limited physical activity and difficult access to healthcare systems.
Therefore, in the present sub-analysis of a study performed in a real-world setting we documented that both empagliflozin and dapagliflozin produce a significant increase in hematocrit and hemoglobin levels in patients with T2DM, regardless of underlying cardiovascular disease at baseline. It is of utmost importance that this increase did not correlate with other established cardiovascular risk factors. According to a recent sub-analysis of the hallmark EMPA-REG OUTCOME trial, change in hematocrit and hemoglobin induced by empagliflozin was the most important mediator of the reduction in heart failure (HF) hospitalization and related death among enrolled subjects with established cardiovascular disease [6], highlighting this indirect effect of SGLT-2 inhibitors and its role in cardiovascular risk reduction. Regarding the association between hematocrit and high-density lipoprotein cholesterol levels, a recent post hoc analysis of 4 randomized controlled trials with empagliflozin led to the conclusion that the observed increase in hematocrit with this SGLT-2 inhibitor is associated with a significant increase in lipid fractions [7].
Despite the significant progress in the clarification of mechanisms underlying the multiple benefits of this drug class [8], it seems that the road to understanding is still wide open. However, their effects are achieved with mechanisms far beyond their glucose-lowering action [9].