Introduction
The atherosclerotic process starts at the tunica intima and tunica media layer in the arterial wall and causes a structural change in the early period as increased intima-media thickness (IMT). Increased IMT of the posterior arterial wall can be detected clearly with ultrasonography (US). In the evaluation of IMT in adulthood the main carotid IMT (CC-IMT) was evaluated with carotid US (C-US) and abdominal aortic IMT (AA-IMT) obtained by abdominal US (A-US) is used to evaluate the signs of atherosclerosis in childhood [1, 2]. IMT is used for early detection of macrovascular complications in both age groups. AA-IMT measurement can be used to detect the development of early atherosclerosis due to the initial onset of atherosclerosis in the distal abdominal aorta [3–5]. There are limited studies regarding the usage of AA-IMT in patients with coronary artery disease (CAD) risk factors [6–11].
However, recently published articles have reported that AA-IMT may be easily measured with high-resolution devices [8–11]. Another important and atherosclerotic process-related IMT measurement region is thoracic aortic IMT (TA-IMT). However, with US devices, this region cannot be evaluated because of its bone structure. TA-IMT measurement with transesophageal echocardiography (TEE) has been performed successfully and objectively for many years, and increased IMT has been associated with advanced atheroma formation, poor prognosis, and early atherosclerosis detection [12–15].
There are many studies which are about AA-IMT and TA-IMT measurements in different patient groups, but there is no study with both AA-IMT and TA-IMT measurements in the same patient group. The aorta has ascending, descending thoracic, and abdominal parts anatomically. The intima layer is seen throughout the aorta. It is still not known which section of the aorta has a higher IMT value and which part of the aortic IMT value is related to CC-IMT.
Our aim was to assess AA-IMT and TA-IMT values obtained by A-US and TEE, respectively, and to investigate their relationship with CC-IMT in patients with CAD risk factors.
Material and methods
Study population
This cross sectional study included 100 patients who underwent TEE examination for different reasons with at least one CAD risk factor. The subjects were divided into two groups as 50 patients with increased CC-IMT (mean age: 52.8 ±7.3 years, male/female: 30/20) and 50 patients with normal CC-IMT values (mean age: 50.2 ±10.1 years, male/female: 37/13). Patients with known CAD or CC, TA and AA atheroma (CC-IMT > 1.5 mm, TA-IMT > 3.00 mm, AA-IMT > 3.00 mm) were excluded (Figure 1). Nineteen patients with known carotid and aortic atheroma or plaque (C-IMT > 1.5 mm) were excluded from the study. Patients with previous aortic operations, secondary or malignant hypertension, pulmonary hypertension, abdominal aortic aneurysm or dissection, congestive heart failure, cerebrovascular disease, moderate-severe valvular heart disease, inflammatory, hematologic diseases, cancer, thyroid disease, moderate-severe liver and kidney disease, pregnancy, active infectious disease, or age under 18 years old were excluded. The Local Ethics Committee approved the study protocol. All patients included in the study were informed about the study and provided their written informed consent.
Detailed physical examination was performed and thorough medical history was taken. Demographic characteristics, systolic and diastolic blood pressure (DBP), and heart rate were recorded. Patients were questioned about the presence of smoking, hypertension, diabetes mellitus, hypercholesterolemia, CAD, and obesity. Body mass index values were calculated. All medications related to CAD risk factors were noted.
Laboratory measurements
Venous blood samples were obtained at polyclinic admission. Samples were taken from the cubital vein into blood tubes. Blood counts were measured by a Sysmex K-1000 (Block Scientific, Bohemia, New York) auto analyzer within 5 min of sampling. Serum glucose, blood urea nitrogen, creatinine, total cholesterol, low-density lipoprotein (LDL) cholesterol, high-density lipoprotein (HDL) cholesterol, triglyceride, high-sensitivity C-reactive protein (hs-CRP) and uric acid concentrations were measured with an automated chemistry analyzer (Abbott Aeroset, Minnesota, USA) using commercial kits (Abbott).
Intima-media thickness measurement of the common carotid artery and abdominal aorta
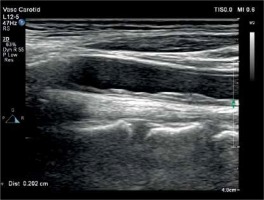
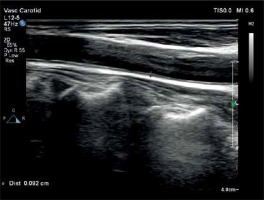
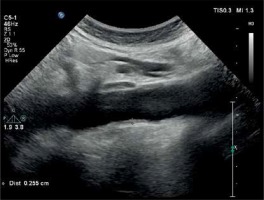
The common carotid artery and abdominal aorta were examined using a high-resolution US device (Philips EPIQ 7) equipped with a linear (12 MHz) and convex (5 MHz) high-resolution transducer (Philips Health Care, Bothell, WA, USA). All arteries were evaluated both longitudinally and transversely. The ultrasound scanner setting was made to be useful for every patient for all B-mode USG examinations (gain 55–75 dB; penetration depth 2.5–16 cm; dynamics range 50–60 and zoom range 0.8–2.0). Analyses were performed on 2 high-quality images. IMT was defined as the distance between front edges of the first and second echogenic lines. The first line represents the intima and lumen border and the second line represents the collagen layer of the adventitia. Two independent and blind observers measured vascular IMTs. All IMT values were calculated as the average of 6 measurements. Patients were in a supine position, and they turned their head to the 45° opposite direction of the examined carotid artery. CC-IMT was measured from the posterior wall of the carotid artery’s pre-bifurcation segment (10–20 mm) (Figure 2). A-IMT was examined at the segment between the renal artery and iliac artery bifurcation. Also, the posterior wall of the abdominal aorta was used for the AA-IMT measurement (Figure 3). CC-IMT values higher than > 0.9 mm were accepted as increased [16].
Intima-media thickness measurement of the descending thoracic aorta
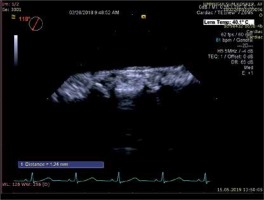
Transesophageal echocardiography examinations were performed with the EPIQ 7 (Philips Healthcare Andover MA, USA). This procedure was applied after 8 h of fasting and the patient was deeply sedated by an anesthesiologist. The TEE probe was a multiplane transesophageal transducer. Patients were positioned in the left lateral decubitus position. The probe was engaged to the esophageal and gastric entry level. The experienced cardiology specialists were blind regarding patient details. All patients well tolerated the TEE procedure and there was no complication. TA-IMT was measured from 6 segments of the thoracic aorta. These segments were: 1) ascending aorta, 2) arch, 3) from 0 to 5 cm distal to the arch, 4) from 5 to 10 cm distal to the arch, 5) from 10 to 15 cm distal to the arch, and 6) from 15 to 20 cm distal to the arch. Mean of the 12 measurements (2 for any segment) was accepted as TA-IMT [15]. TA-IMT was defined as the distance from the leading edge of the lumen-intima interface to the leading edge of the media-adventitia interface of the posterior or far wall (Figure 4).
Statistical analysis
All analyses were done in SPSS 22.0 (IBM SPSS Statistics Version 22.0. Armonk, NY: IBM Corp.) software. Continuous variables and categorical variables were expressed as mean ± standard deviation and numbers and percentages, respectively. The k coefficient was used to examine the interobserver variability of all the IMT measurements. The Kolmogorov-Smirnov test was used to test the normality of continuous variables. Student’s t test was used for the normal distributed variables. The Mann-Whitney test was used for the variables without normal distribution. Categorical variables were analyzed by the chi-square (χ2) test. Multivariate linear regression analysis was performed to evaluate the CC-IMT and AA-IMT relationship. Also, logistic regression analysis was used to detect the parameters which independently determine the patients with CC-IMT > 0.9 mm. Receiver operating curve (ROC) analysis was done. P < 0.05 was accepted as statistically significant.
Results
AA-IMT and TA-IMT measurements were successfully obtained from all patients included in the study after exclusion criteria. Mean, median, minimum and maximum AA-IMT values of all patients were 1.55 ±0.27, 1.60, 1.00 and 3.00 mm, respectively. Mean, median, minimum and maximum TA-IMT values of all patients were 1.39 ±0.25, 1.38, 0.85 and 2.50 mm, respectively. Mean, median, minimum and maximum Δ aortic IMT values (difference between AA-IMT and TA-IMT) of all patients were 0.16 ±0.15, 0.14, –0.17 and 0.53 mm, respectively. Cohen k values that evaluate interobserver and intraobserver variability were over 90% for all IMT measurements. AA-IMT values were higher than TA-IMT in all patients except 11 patients. The study population was divided into two groups as increased CC-IMT (CC-IMT > 0.9 mm) and normal CC-IMT (CC-IMT (0.9) according to CC-IMT values and all parameters were compared. Demographic, clinical, laboratory and IMT values of study groups were compared. When the demographic and clinical data of both groups were evaluated, the prevalence of hypercholesterolemia and HT, SBP and DBP was found to be higher in patients with increased CC-IMT, and other demographic and clinical parameters were similar between the two groups (Table I).
Table I
Demographic, clinic and laboratory findings in patients with increased and normal carotid intima-media thickness
Creatinine, total cholesterol, LDL cholesterol, triglyceride, hs-CRP, uric acid, CC-IMT, AA-IMT, and TA-IMT values were significantly higher and HDL cholesterol levels were significantly lower in patients with increased CC-IMT (Table I).
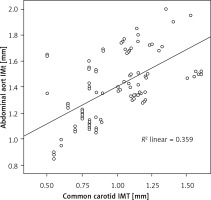
AA-IMT and TA-IMT values were significantly higher in patients with increased CC-IMT (Table II). In the logistic regression analyses AA-IMT, systolic blood pressure, total and HDL cholesterol levels independently determined the patients with increased CC-IMT (CC-IMT > 0.9 mm). According to this analysis, it was found that every 0.1 mm increase in AA-IMT value doubled the probability of having increased CC-IMT (Table III). In addition, linear regression analysis showed that CC-IMT values were closely and independently related to AA-IMT (p < 0.001 and β = 0.599, Figure 5).
Table II
Vascular ultrasound findings in patients with increased and normal carotid intima-media thickness
Table III
According to multivariate regression analysis, independent risk factors for occurrence of increased carotid intima-media thickness
Figure 5
Simple scatter/dot diagram for the relationship between the main carotid and thoracic aortic intima-media thickness: a close relationship between the main carotid and thoracic aortic intima- media thickness
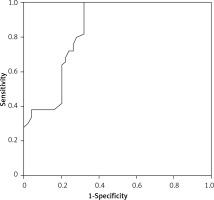
ROC curve analysis was also performed to identify patients with CC-IMT > 0.9 mm; the parameters AUC > 0.700 are shown in Table IV. The area under the ROC curve was maximum for AA-IMT and the area under the curve was found to be 0.843, 0.785 and 0.757 for AA-IMT, TA-IMT and SBP respectively. A cut-off value of 1.5 mm for AA-IMT detected increased CC-IMT with 86% sensitivity and 82% specificity (Figure 6).
Table IV
ROC curve analysis of abdominal aort intima-media thickness and office systolic blood pressure for predicting increased carotid intima-media thickness
Discussion
The most important finding of our study was that the AA-IMT value was significantly higher than TA-IMT in patients with CAD risk factors. To the best of our knowledge, this was the first study in which AA-IMT and TA-IMT were determined in the same patient group at the same time. Another important finding of our study was that the AA-IMT value was significantly and more closely related to increased CC-IMT than the TA-IMT value. When the AA-IMT cut-off value was taken as 1.5 mm, it determined the patients with increased CC-IMT with acceptable sensitivity and specificity.
CAD risk factors: i) increase smooth muscle cell proliferation in the vessels, ii) cause oxidative stress in tissues, iii) thicken the intima-media layer of the vessel with increasing free oxygen radicals [2, 16–19]. Regular CC-IMT evaluation is advised to detect the subclinical atherosclerosis in patients with CV risk factors such as hypertension, diabetes, hyperlipidemia, smoking, advanced age, and obesity [16–19]. There are many studies which have shown a close relation between CC-IMT and CAD, myocardial infarction, and stroke. Therefore, CC-IMT evaluation is advised as a routine screening method [13, 16–19]. Carotid artery evaluation is a preferred method, because it is superficially located, easy to find and easy to visualize. TA-IMT can be easily measured during the TEE evaluation, and it can be used for detection of early atherosclerosis and as a follow-up method for CV diseases [12–15]. However, TEE is invasive and it can be used in a limited number of patients; it cannot be used in routine practice of patients with CV risk factors. Moreover, it only can be useful for a limited patient group who need an evaluation with TEE. The abdominal aorta is another important IMT measurement location. It is a location in which the first atherosclerotic lesions and early signs of atherosclerosis such as fatty streaks can be seen. A limited number of studies have mentioned the importance of AA-IMT as a screening method for detection of early atherosclerosis in adults [6–11, 14, 15]. Moreover, the abdominal aorta is the first area to be affected by atherosclerosis in pediatric patients with CV risk factors. So, AA-IMT is the first choice in childhood. The main limiting factors for AA-IMT evaluation are abdominal fat tissue and usage of low penetrating US devices [6]. However, it is shown that new high-resolution US devices and probes made possible reliable AA-IMT measurements [8–11].
Due to these limitations and difficulties in the measurement of AA-IMT and TA-IMT, we did not find any study comparing the TA-IMT and AA-IMT values in the same patient group and measuring IMT from both regions in the literature. In our study IMT was measured from both aortic anatomical regions for the first time. We found that AA-IMT values were higher than TA-IMT values. This finding supports the hypothesis that an increase in vascular IMT was initially initiated from AA. Another important finding was the comparison of CC-IMT ant the IMT values obtained from the TA and AA. AA-IMT values were more closely related to CC-IMT. It was shown that AA-IMT could independently predict the patients with increased CC-IMT. Every 0.1 mm increase in AA-IMT value doubled the probability of having increased CC-IMT (Table III). A cut-off value of 1.5 mm for AA-IMT was shown to be potentially useful to detect patients with increased CC-IMT. Recent studies have also reported that a 1.5 mm cut-off value could be used to document increased AA-IMT in patients with hypertension and diabetes mellitus [9, 10]. Abdominal US is a visualization method which can be used in many CV diseases. AA-IMT may have a future like CC-IMT as a screening method to detect early atherosclerosis or subclinical organ damage in patients with CV risk factors.
There are some important limitations of this study. We investigated the relation between AA-IMT and TA-IMT in a cross-sectional study design. Our patient size was relatively small; nevertheless, we showed that there was a significant relation between increased AA-IMT and increased CC-IMT. The study could be more meaningful if it was done prospectively and with more patients. We excluded patients with known CAD and presence of atheroma in any vascular regions. Some of the patients included in our study have been taking medications which could have effects on IMT. This may have affected our results. A recent study reported that age could be very closely related to CC-IMT and therefore a limit value for CC-IMT should be specified according to age [20]. There is no study about A-IMT measurement but the C-IMT measurement can be measured automatically and semi-automatically with new software programs, resulting in a lower average value than the manual measurement [21, 22]. This automatic measurement especially removes operator dependence and is more useful for repetitive measurements. However, our high-resolution device did not have this software program so we could not make this evaluation. In our study, if IMT could be measured automatically and semi-automatically, more objective and meaningful results could be obtained. We used only 0.9 mm as a cut-off value in all patients. In addition, our study was not a follow-up study and did not evaluate the prognosis of CV diseases. We used high-resolution US devices and probes. Therefore, it may not be possible to achieve similar results with low frequency and low-resolution devices.
In conclusion, AA-IMT values are higher than TA-IMT in patients with different CAD risk factors. Also AA-IMT is more closely related to CC-IMT than TA-IMT. According to our results, patients with an AA-IMT value > 1.5 mm should be closely followed. CC-IMT measurement is advised in hypertension, diabetes, and hyperlipidemia treatment guidelines. AA-IMT should also be a part of A-US, which is a screening method for these diseases, and AA-IMT values should be reported in the A-US results.