Introduction
Millions of individuals worldwide suffer from atrial fibrillation (AFib), a common arrhythmia that is usually associated with increased morbidity and mortality from thromboembolic events, e.g., stroke [1]. This disease is a supraventricular tachyarrhythmia characterized by uncoordinated atrial activation with ineffective atrial contraction, increasing the risk of stroke and thromboembolism due to thrombus formation, typically in the left atrial appendage (LAA) [2]. Thus, a critical component of addressing AFib is adequate treatment with anticoagulation, which reduces the risk of stroke and systemic embolism [3].
For multiple years, the cornerstone of anticoagulant therapy for patients with AFib has been the vitamin K antagonist (warfarin). However, the introduction of direct oral anticoagulants (DOAC) has completely changed the landscape, providing a warfarin alternative with several advantages, including lower drug-drug interactions and no need for regular monitoring plus far more predictable pharmacokinetics [4].
Chronic liver disease (CLD) places a significant burden on global health. Coagulopathy is a common symptom in CLD patients, which makes managing anticoagulation requiring disorders like AFib, more difficult [5]. Patients with CLD often have a malfunctioning hepatic synthetic framework, which naturally prolongs prothrombin time (PT) and may increase the bleeding risk. This leads to lower levels of clotting factors [6].
Although warfarin is a widely prescribed anticoagulant, patients with chronic liver disease (CLD) face significant challenges when using it. Warfarin is primarily metabolized by hepatic cytochrome P450 enzymes, meaning liver function greatly influences its metabolism. This variability leads to fluctuations in the international normalized ratio (INR), necessitating frequent monitoring and dose adjustments [7]. Warfarin’s narrow therapeutic window and numerous drug-food interactions complicate treatment for CLD patients, who often take multiple medications. Additionally, warfarin’s antagonistic effect on vitamin K may exacerbate underlying coagulopathy, increasing the risk of severe bleeding events [8].
For patients with non-valvular AFib, DOAC such as dabigatran, rivaroxaban, apixaban, and edoxaban have emerged as effective alternatives to warfarin. These medications offer more predictable pharmacokinetics and specifically target clotting factors – thrombin for dabigatran and factor Xa for the others – allowing for fixed dosing without the need for frequent monitoring [9]. However, concerns remain regarding their safety and efficacy in patients with hepatic impairment as the liver only metabolizes a part of DOAC [10].
Consequently, current guidelines provide limited recommendations for their use in CLD patients, forcing physicians to navigate treatment decisions with minimal information. Clinicians must weigh the risks of thrombosis against the potential for severe bleeding, requiring a thorough assessment of the patient’s liver disease severity, bleeding risk, and possible drug interactions. Patients with both AF and CLD face difficult choices between warfarin and DOAC, highlighting the need for careful consideration in their management [11].
Given the significant risk of both severe bleeding and thrombosis, a full analysis of the available evidence on the comparative safety of DOAC vs. warfarin in patients with AFib and CLD is of paramount importance. The main goal of this systematic review is to provide a thorough analysis of the body of research that has already been done, with a focus on the incidence of serious bleeding.
Analytical techniques and materials
Research retrieval strategy
Cochrane methodological criteria were followed while writing this meta-analysis, and the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines served as the basis for reporting the findings [12].
A comprehensive review of the literature was conducted in the PubMed, Medline, and the Cochrane Library databases, covering the period from 1980 to August 2024, to identify trials comparing warfarin with DOAC in individuals with CLD and AFib, as well as previously published meta-analyses on the same topic.
We used the following keywords:
(“direct oral anticoagulants” OR “Oral Factor Xa Inhibitors/administration and dosage” OR “non-vitamin K antagonist oral anticoagulant”)
AND (warfarin OR “Warfarin” OR “vitamin K antagonist oral anticoagulant”) AND (“atrial fibrillation” OR “Atrial Fibrillation” OR “AF”) AND (“liver disease” OR “chronic liver disease” OR “Liver Cirrhosis” OR “End Stage Liver Disease” OR “liver dysfunction”). Detailed search strategy is shown in Supplementary Table SI.
Zotero was utilized to manage the screening process, including the organization of references, removal of duplicates, and tracking of the selection criteria. Additionally, a manual search of reference lists from relevant studies, reviews, and meta-analyses was conducted.
Inclusion and exclusion criteria
The inclusion criteria for this study required adult patients diagnosed with atrial fibrillation (AFib) and chronic liver disease (CLD). Eligible studies included randomized clinical trials, case-control studies, and cohort studies that reported risk estimates with 95% confidence intervals (CIs) and provided sufficient data for these calculations. Additionally, studies had to involve patients with AFib and liver disease, with outcomes measured over a minimum follow-up of 1 month. Only studies involving patients receiving DOAC, with warfarin as a comparator, were included. Exclusions comprised meeting abstracts, reviews, editorials, letters, commentaries, animal studies, studies on valvular AFib, those with duplicate data to ensure the analysis’s integrity, and studies with vitamin K antagonists other than warfarin.
Outcomes
The primary outcomes of this meta-analysis were all-cause death, ischemic stroke/systemic embolism and major bleeding. The secondary outcomes were major GI bleeding, all-cause bleeding, intracranial haemorrhage, and ischemic stroke.
Data extraction
To ensure data accuracy and minimize bias, standardized procedures guided the data extraction process. A pilot-tested data extraction form was developed to collect relevant information from included studies. Two independent reviewers screened titles and abstracts, followed by full-text assessments of potentially eligible studies. Disagreements were resolved by a third reviewer.
The data extraction form captured the following key information: Study characteristics, Intervention and comparator details, Outcome data and Other relevant data.
Bias assessment and outcome validity
The methodological quality of included observational studies was assessed using the ROBINS-I (Risk Of Bias In Non-randomized Studies – of Interventions) tool. The overall risk of bias for each study was determined by the highest risk level identified in any of the seven domains. For instance, if a study was rated as having a ‘serious’ risk of bias in one domain but ‘low’ or ‘moderate’ in others, it was classified as having an overall ‘serious’ risk of bias. To visually summarize the risk of bias assessments, we used the ROBINS-I tool to generate traffic light plots.
Consideration of intention-to-treat analysis
We noted that five of the included studies employed an intention-to-treat (ITT) approach. These studies are: Lawal [13], Lee [14], Wang [15], Qamar [16], and Serper M 2021. The use of ITT was considered when assessing the risk of bias, particularly in the domains related to deviations from intended interventions and missing data. The identified studies were assessed for bias using the Cochrane Collaboration’s criteria [17].
Statistical analysis
Review Manager version 5.4 was used for statistical analysis. For each study included, the chosen impact measurement estimates were hazard ratios (HRs) along with their corresponding 95% CIs. The effect estimates were computed using the total number of patients and events, if available. Statistical software was used to determine the natural logarithm of the HR (log [HR]) and its standard error (SElog [HR]), which were then pooled. The heterogeneity for treatment effects was evaluated using I2 values, with 25–50%, 50–75%, and ≥ 75% representing moderate, intermediate, and severe heterogeneity, respectively. Using a random-effects model, we arrive at a rather conservative conclusion. The difference in population (Asian vs. non-Asian) was the basis for the subgroup analysis. A total of nine studies were included for systemic review but only seven studies were included in the analysis excluding Pastori et al. [18] and Yoo et al. The study by Pastori et al. [18] was included in the review but excluded from the analysis due to its different data format. While other studies report hazard ratios (HRs) and confidence intervals, the Pastori et al. study utilizes the FIB-4 score to classify liver fibrosis and links these scores to bleeding events without providing directly comparable HRs. It discusses bleeding risks through event rates in various patient groups instead of using HRs needed for meta-analysis. Similarly, the study by Yoo et al. was excluded because it does not present HRs comparing bleeding events between DOAC and warfarin, despite using a Cox model and reporting cumulative bleeding rates.
Results
Search results
The search identified a total of 74 articles. After removing 5 duplicates with Zotero, 45 articles were excluded based on the title and abstract review. Of the remaining 23 articles, 3 were found to meet the inclusion criteria. Studies were excluded if they were reviews, comments, did not meet the population criteria, or had missing data or irrelevant outcomes. By adding 6 studies from a previously published systematic review on the same topic [19], a total of 9 studies were included in this updated review. A manual search of reference lists from relevant studies, reviews, and meta-analyses did not uncover any additional eligible studies. The PRISMA diagram in Figure 1 illustrates the comprehensive search strategy employed.
Baseline characteristics
This comprehensive analysis involved 53,224 participants, divided into two groups: DOAC and Warfarin. With 62.59% of individuals in the DOAC group and 37.40% of individuals in the warfarin group. Detailed information about the study characteristics and the baseline characteristics of the participants can be found in Table I.
Table I
Baseline characteristics
Risk of bias assessment
Our assessments indicated variability among the studies. Notably, two studies (Goriacko [20] and Serper 2021) were identified as having a serious overall risk of bias, primarily due to confounding issues and missing data. The other studies exhibited a moderate overall risk of bias, with common problems related to confounding, participant selection, and missing data. The risk of bias across seven domains is illustrated in traffic light plots and a weighted bar plot (see Supplementary Figures S1 and S2). Sensitivity analyses for primary outcomes examined the impact of studies with serious bias on overall findings. We concluded there is a moderate overall risk of bias.
All-cause death
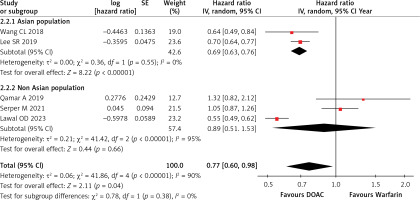
Five studies reported on all-cause mortality. The combined analysis indicated a statistically significant difference between the two groups, with the DOAC arm showing a 23% reduced risk of all-cause death compared to the warfarin group (HR = 0.77; 95% CI = 0.60–0.98; I2 = 90%; p = 0.04), as illustrated in Supplementary Figure S3. Subgroup analysis revealed a significant reduction in all-cause death risk in the Asian population (HR = 0.69; 95% CI = 0.63–0.76; I2 = 0%; p < 0.00001) using DOAC, while no significant reduction was observed in non-Asians (HR = 0.89; 95% CI = 0.51–1.53; I2 = 95%; p = 0.66), as shown in Figure 2. A leave-one-out sensitivity analysis was performed due to high heterogeneity, but no significant differences were noted after removing individual studies, as shown in Supplementary Table SII.
Ischemic stroke or systemic embolism (ISSE)
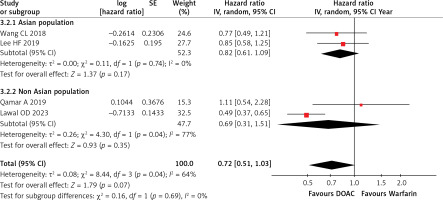
Four studies included in the analysis reported cases of ISSE. The combined data indicated a reduced ISSE risk with DOAC compared to warfarin (HR = 0.72; 95% CI = 0.51–1.03; I2 = 64%; p = 0.07), though this was not statistically significant (Supplementary Figure S4). Subgroup analyses showed no significant reduction in ISSE risk for Asians (HR = 0.82; 95% CI = 0.61–1.09; I2 = 0%; p = 0.17) or non-Asians (HR = 0.69; 95% CI = 0.311.51; I2 = 77%; p = 0.35), as shown in Figure 3. A leave-one-out sensitivity analysis revealed that excluding Qamar 2019 [16] resulted in significant findings (HR = 0.67; 95% CI = 0.46–0.97; I2 = 68%; p = 0.03), while removing Lawal 2023 [13] decreased heterogeneity (HR = 0.85; 95% CI = 0.65–1.12; I2 = 0%; p = 0.25), as shown in Supplementary Table SIII.
Major bleeding
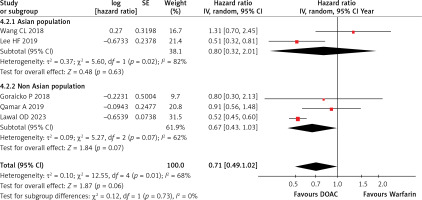
Five studies reported major bleeding events. The analysis indicated that there was reduced bleeding with DOAC compared to warfarin, but it was not statistically significant (HR = 0.71; 95% CI = 0.49–1.02; I2 = 68%; p = 0.06), as illustrated in Supplementary Figure S5. Subgroup analysis found no significant reduction for Asians (HR = 0.80) or non-Asians (HR = 0.67), as illustrated in Figure 4. A leave-one-out sensitivity analysis showed significant results when removing Wang 2018 [15] (HR = 0.60; 95% CI = 0.45–0.79; I2 = 44%; p = 0.0003), as shown in Supplementary Table SIV.
Major GI bleeding
Four included studies reported major gastrointestinal (GI) bleeding. Combined analysis of the data revealed a statistically significant difference between the two groups. There was a slight reduction in major GI bleeding with DOAC compared to the warfarin group (HR = 0.87; 95% CI = 0.49–1.53; I2 = 82%; p = 0.62) as shown in Table II.
All-cause bleeding
Out of the seven studies, three specifically reported on all-cause bleeding. The combined analysis of the data revealed a statistically insignificant difference between the two groups. The pooled analysis indicated a trend towards a reduction in all-cause bleeding with DOAC compared to the warfarin group (HR = 0.78; 95% CI = 0.46–1.32, I2 = 70%; p = 0.35), as shown in Table II.
Intracranial haemorrhage
Out of the seven studies, three reported on the risk of intracranial haemorrhage. The combined analysis of the data revealed a statistically significant difference between the two groups. The DOAC arm was associated with a significantly lower risk of intracranial haemorrhage compared to the warfarin group (HR = 0.49; 95% CI = 0.40-0.59, I2 = 0%; p < 0.00001), as shown in Table II.
Ischemic stroke
Three studies out of the included seven compared the risk of ischemic stroke between DOAC and warfarin. The pooled hazard ratio (HR = 0.63; 95%CI = 0.32–1.22, I2 = 68%; p = 0.17) suggests a trend toward a reduction in ischemic stroke risk with DOAC compared to warfarin. The overall effect is not statistically significant as shown in Table II.
Discussion
The results of the updated meta-analysis comparing the efficacy of DOAC with that of warfarin in patients with CLD indicated that DOAC were associated with significantly decreased risks of all-cause death and intracranial haemorrhage. These results are in line with the findings of the previous meta-analyses that reported similar effect sizes.
In AFib patients with liver disease, the imbalance in pro- and anti-coagulation factors (due to the liver dysfunction) acts as a risk factor in anticoagulation therapy and can precipitate either excessive bleeding (leading to ICH) [21] or thromboembolism (leading to stroke and death) [22]. Anticoagulation therapy is essential in preventing both embolization and haemorrhage in patients with liver dysfunction. Vitamin K antagonists (VKAs) prevent the activation of clotting factors by inhibiting gamma-carboxylation of glutamic acid residues, while direct oral anticoagulants (DOAC) inhibit clotting factors IIa and Xa after their production [23].
One likely reason for the reduced risk of all-cause death and intracranial haemorrhage (ICH) with DOAC is that they directly inhibit two specific clotting factors (II and Xa), while warfarin indirectly targets multiple vitamin K-dependent factors, resulting in variability in its anticoagulant effect. Additionally, warfarin’s longer half-life means it can take days to restore normal clotting levels after discontinuation, increasing ICH risk. In contrast, DOAC have a shorter half-life, allowing their effects to dissipate more quickly in the event of bleeding.
Furthermore, warfarin’s numerous food and drug interactions make maintaining stable therapeutic levels challenging. Therefore, DOAC may offer a more effective and reliable anticoagulant therapy with fewer interactions and monitoring requirements. Several studies supported this evidence [13–16, 18, 20, 22, 23]. Another significant issue with anticoagulants in liver disease is that several of them are metabolized via hepatic CYP enzymes, including warfarin, apixaban, and rivaroxaban. With warfarin, impaired metabolism would become evident through monitoring the INR. However, apixaban and rivaroxaban are not routinely monitored via coagulation testing or assay, meaning the effect of impaired metabolism may not be quantified or even recognized [24].
As seen in the forest plots generated by the random-effects model, the two statistically significant outcomes whose risks are decreased by the usage of DOAC were all-cause death and intracranial haemorrhage. There was no heterogeneity observed in the results of intracranial haemorrhage (0%), whereas the heterogeneity observed for all-cause death was significantly high (90%) and may be due to differences in study populations.
We performed the subgroup analysis for all-cause death based on the study populations, i.e. Asian and non-Asian patients. The pooled analysis showed that treatment with DOAC was significantly associated with a decreased risk of all-cause death, while on subgroup analysis, it was shown that in Asian populations, the risk of all-cause death was significantly reduced (p = 0.00001) as compared to non-Asian populations (p = 0.66). In terms of heterogeneity, it was 0% across all (Asian and non-Asian) studies for the ICH outcome, but in case of the all-cause death outcome, it was 0% for Asian patients (indicating consistencies across all studies) and 95% for non-Asian patients (indicating significant variability across all studies).
Outcomes such as all-cause bleeding, ischemic stroke, systemic embolism and gastrointestinal bleeding had heterogeneities ranging from 64% to 82% and all were statistically insignificant. It must be noted that the risk for gastrointestinal bleeding in cirrhotic patients may significantly reduce through the usage of proton pump inhibitors and lead to further heterogeneity. However, there was only limited evidence on this as there were only two studies that reported specific data on PPIs [13, 22].
The validity of our meta-analysis is subject to several limitations. First, the definitions of liver disease varied among the included studies, and quantitative measures such as Child-Pugh scores were provided in only a few, contributing to potential heterogeneity. Second, all studies analysed were either observational or post hoc analyses of randomized controlled trials (RCTs), increasing the likelihood of bias. Although sensitivity and subgroup analyses were performed to mitigate these issues, some limitations remain inherent to these methodological designs. Third, genetic polymorphisms affecting warfarin metabolism can lead to differences in efficacy and INR data among patients, which may explain the superior outcomes associated with DOAC. Fourth, the findings of this study are based on observational data and should be interpreted with caution until they are validated through randomized controlled trials. Another significant limitation is the inconsistent definition of major bleeding events across studies. While three studies adhered to the International Society on Thrombosis and Haemostasis (ISTH) criteria, Wang et al. [15] defined major bleeding as hospitalization for bleeding and blood transfusion within 14 days of drug use, potentially skewing major bleeding event rates. Generalizing our findings to patients with multiple comorbidities or limited healthcare access may also be challenging.
In conclusion, this was an updated meta-analysis that compared the effect of DOAC against warfarin for anticoagulation therapy in patients of atrial fibrillation with concomitant liver disease. The analysis found that, compared to warfarin, DOAC were associated with lower risk of ICH among patients with atrial fibrillation and liver disease. A lower risk of all-cause mortality was also observed within Asian populations treated with DOAC vs. warfarin.