Cardiovascular disease morbidity and mortality are recognized to be increased by raised low-density lipoprotein (LDL) in hypercholesterolaemia such as familial hypercholesterolaemia (FH) [1, 2]. For patients with cardiovascular disease who are at risk, 3-hydroxy-3-methylglutarylcoenzyme A reductase inhibitors, statins, are the most commonly used medications to reduce LDL cholesterol levels [3]. However, there are still unresolved issues regarding residual risk factors beyond LDL cholesterol that need further research [1, 2].
While high-density lipoprotein (HDL) itself is thought to account for one of the residual risk factors, paraoxonase-1 (PON1), an enzyme carried by high-density lipoprotein (HDL), is known to be essential for the antioxidant activity of HDL to prevent cardiovascular disease [4, 5]. PON1 reduces LDL oxidation by the hydrolysis of lactones, providing a molecular insight into the prevention of cardiovascular disease [4, 5].
In a previous study we showed that PON1 lactonase activity was higher in patients with FH than in controls [6]. This seemed surprising and counterintuitive because disease-prone patients were generally reported to have lower PON1 activities when measured as arylesterase and paraoxonase (PON1 is a promiscuous enzyme, and the substrates typically used for measurements are not lactones) [7]. Because of this, we thought it would be interesting to examine the changes of PON1 lactonase activity after statin treatment to account for the high lactonase activity [6].
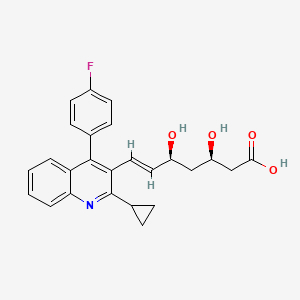
In total, 12 patients with defined FH and 6 with probable FH made up the studied population (n = 18, mean age: 60 years, men: 39%). These patients were clinically diagnosed due to the presence of hyper-LDL-cholesterolaemia, xanthomas, and/or a family history of premature coronary artery disease in accordance with the guidelines [8]. Included were patients older than 40 years who did not have obvious heart disease. Those who were non-compliant and had a statin intolerance were excluded. Pitavastatin [9] (n = 16: 1 mg/day, n = 2: 2 mg/day) was administered to the patients for a duration of 3 months. The levels of LDL-cholesterol, HDL-cholesterol, and triglycerides were evaluated enzymatically in fasting blood both before and after treatment. Simultaneously, PON1 lactonase activities were kinetically measured in an in-house assay by hydrolysis of 5-(thiobutyl) butyrolactone for product detection using absorbance at 404 nm [10]. The Institutional Research Ethics Committee approved the study, and the patients gave written informed consent.
A paired t-test was used in pre- and post-changes of lipid levels and PON1 lactonase activities. Data are presented as the mean ± standard deviation, except for triglycerides, which are presented as the median (interquartile range) due to their skewed deviation. A p-value of < 0.05 was set as significant.
During the treatment period, the LDL cholesterol levels were significantly reduced (pre: 4.6 ±0.9, post: 3.1 ±0.5 mmol/l; p < 0.01), while the levels of HDL cholesterol (pre: 1.9 ±0.7, post: 1.9 ±0.7 mmol/l; p = 0.24) and triglycerides (pre: 1.1 [0.8–2.1], post: 1.2 [0.8–2.0] mmol/l; p = 0.61) were unchanged. PON1 lactonase activities were significantly reduced with treatment (pre: 70 ±22, post: 67 ±20 U/l; p < 0.01).
In the current study, we observed the reduction of PON1 lactonase activity after pitavastatin treatment in this population. While there is scarce information available on lactonase activity, statin treatment is often revealed to boost PON1 arylesterase and paraoxonase activities [11]. The rise in PON1 is assumed to result from various factors, such as the statin-induced PON1 expression in hepatocytes or the pleiotropic antioxidative effects of statins, which preserve PON1 while reducing LDL oxidation [11]. We hypothesized that the high PON1 lactonase activity might be the result of a compensatory response to oxidative burden, as seen in FH in our previous report [6]. Then, the pleiotropic antioxidative action of statin may render such a compensatory reaction unnecessary, leading to a reduction of lactonase activity. To better understand the pathophysiology of the PON1-statin relationship and the roles played by PON1 species in cardiovascular protection, more research should be done on this conjecture.
There are limitations to the current investigation. It was a small-sampled, short-term, and single-arm study. The other PON1 species were not comparatively examined. Although PON1 activities can be modified by several factors (e.g. diet [10]), lifestyle factors were not monitored during the treatment period. Although the effects of statins on PON1 activities are believed to be the same regardless of statin type, dose, treatment duration, and LDL-cholesterol changes [11], there is scarce data on PON1 activities with pitavastatin, and comparative studies with other statins may be needed. These must be addressed in future work.
In conclusion, PON1 lactonase activities were reduced after pitavastatin treatment in patients with hypercholesterolaemia. Further studies are required.