The aging of society is progressing in developed countries, and sarcopenia is recognized as an age-related entity that manifests a low muscle mass with muscle dysfunction [1]. Of note, not only musculoskeletal disorders but also atherosclerotic cardiovascular diseases occur in sarcopenia [1, 2]. Although sarcopenia has multifactorial causes and its pathophysiology is not fully understood, oxidative stress is known to be involved in muscle damage detected in sarcopenia [3, 4].
With regard to a link between sarcopenia and atherosclerotic cardiovascular diseases, lipoprotein oxidation is considered to be possibly involved. In fact, an epidemiological study previously reported that low-density lipoprotein (LDL) oxidation predicted mobility limitation, and a high level of it was able to accurately predict severe mobility limitation [5]. However, other than that previous study [5], we do not know of any other study that has investigated lipoprotein oxidation in sarcopenia. In particular, as high-density lipoprotein (HDL) affects not only atherosclerotic formation but also muscle cell metabolism [6], a potential relationship between HDL oxidation and sarcopenia is of concern. We therefore performed a pilot study that compared the levels of oxidized HDL (oxHDL) between subjects with and without severe sarcopenia, as severe sarcopenia carries a definite risk of mortality [7].
A total of 64 older women (age: 60–89 years old) were enrolled in a clinical setting. We excluded those with acute-phase disease conditions and obvious heart disease. The Institutional Ethics Committee approved the study, and the subjects gave their informed consent. Severe sarcopenia was defined as having low levels of all three criteria: muscle mass of < 5.7 kg/m2 according to a bioimpedance analysis (InBody Inc., Tokyo, Japan), grip strength of < 18 kg according to a dynamometer, and gait speed of < 1.0 m/s according to an accelerometer, as described in the guideline [2]. The existence of diabetes mellitus was based on the medical history. In fasted serum samples, the HDL and LDL cholesterol levels were enzymatically determined, and the oxHDL levels were measured using an enzyme-linked immunosorbent assay (Hoken-Kagaku West, Co. Ltd., Kyoto, Japan) [8, 9]. A t-test and χ2 test were conducted between the groups, as well as a general linear model analysis for the oxHDL levels with adjustment for variables. p < 0.05 was considered significant.
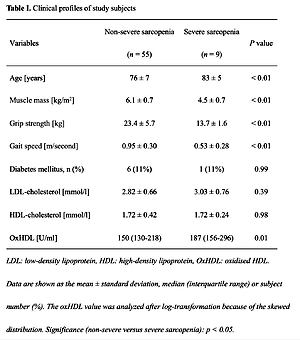
Table I shows the clinical profile of the study population, which included nine subjects with severe sarcopenia. The subjects with severe sarcopenia showed a lower muscle mass, lower grip strength and slower gait speed than those with non-severe sarcopenia. The subjects with severe sarcopenia also had a higher level of oxHDL and a similar level of HDL cholesterol compared to those with non-severe sarcopenia. The difference in oxHDL levels between subjects with severe and non-severe sarcopenia remained significant even after adjusting for age, existence of diabetes mellitus and LDL and HDL cholesterol values (oxHDL, p = 0.04).
Table I
Clinical profiles of study subjects
[i] LDL – low-density lipoprotein, HDL – high-density lipoprotein, OxHDL – oxidised HDL. Data are shown as the mean ± standard deviation, median (interquartile range) or subject number (%). The oxHDL value was analysed after log transformation because of the skewed distribution. Significance (non-severe versus severe sarcopenia): p < 0.05.
Thus, this study revealed the presence of a high oxHDL level in subjects with severe sarcopenia, a finding that was independent of the HDL cholesterol level. While the reasons for the results are unclear at present, the data regarding HDL oxidation may partly account for the link between sarcopenia and atherosclerotic cardiovascular diseases. First, as one of the various oxidative stress markers, oxHDL may simply increase under the excess oxidative burdens which are often seen in severe sarcopenia. However, such oxidative stress conditions generally lead to the development of atherosclerosis [5], thereby resulting in the onset of cardiovascular diseases. Second, from the perspective of the HDL function, oxHDL can show a deficit of anti-atherothrombotic properties, even in cases with normal HDL cholesterol levels [8, 9], thereby leading to cardiovascular diseases. Oxidative stress is known to induce changes in the HDL particles in terms of structure, size, density and composition with altered activities of HDL-associated molecules such as paraoxonase 1 [10, 11]. This merits further mechanistic investigations.
Several limitations associated with the present study warrant mention. The study population was relatively small and restricted to women. Healthy controls were not prepared for analysis. That said, as a pilot study, we obtained seemingly useful results that promote further exploration of this topic. We acknowledge that the cross-sectional design failed to conclusively demonstrate causality. There are many contributors to sarcopenia, and confounders (e.g. lifestyle habits) were not completely regulated. These points will be addressed in the future.
In conclusion, a high level of oxHDL was observed in subjects with severe sarcopenia. This may suggest the potential usefulness of biomarkers related to HDL dysfunction to elucidate the link between sarcopenia and atherosclerotic cardiovascular diseases. More studies are warranted.