Introduction
Aortoenteric fistulas were first described by Cooper [1] as a complication of abdominal aortic aneurysms, while the first case is described in 1843 by Salmon [2] and defined as the abnormal communication between the aorta and the gastrointestinal tract. Aortoenteric fistulas are divided into primary and secondary usually after abdominal aortic aneurysm recovery and are a rare but quite dangerous cause of gastrointestinal bleeding that the general surgeon may face during his/her career. Secondary aortoenteric fistula (SAEF) was first described by Brock in 1953 in the case of a 44-year-old woman 3 months after AAA operation [3]. This review presents the role of the surgeon in the management of secondary aortoenteric fistulas.
The aim of our review is to provide a brief overview of a rare but potentially rapidly fatal condition that general surgeons worldwide might be called to face in the emergency setting. Although specialised management of this condition is paramount to the effective treatment of the SAEF, emergency presentations often require management and surgically-led stabilisation from the general surgeon that is not trained in vascular surgery.
We summarised, in a concise way, the knowledge deriving from the available literature about SAEFs, providing the surgeon’s view.
Material and methods
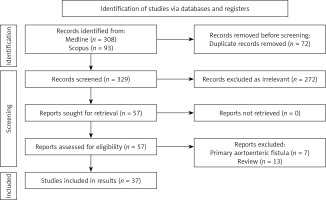
Studies were identified by searching electronic databases and scanning bibliographic references of articles. The National Library of Medicine’s Medline database was searched using the PubMed interface and Scopus from 1990 to 2023. No language constraints were used. The last search was run on 12 December 2023. Keywords were selected using medical subject headings (MeSH) for PubMed and MeSH/Emtree for Scopus. The keywords were “secondary fistula”, “aortoenteric”, “aortojejunal”, and “aortoiliac”. The databases were searched with an unrestricted search strategy, applying exploded MeSH and keywords combined with the Boolean operators AND or OR to retrieve relevant reports as reported in Table I. A second-level search included a manual screen of the reference lists of the articles identified through the electronic search. Eligibility assessment was performed independently in an unblinded, standardised manner by 2 reviewers; disagreements between reviewers were resolved by consensus (Figure 1).
Results
Epidemiology
SAEFs can occur after both open and intravascular reconstructive surgery for an aortic aneurysm or other aortic diseases with incidence ranging from 0.36 to 1.6% and increasing to 12–33% in cases who have an infected graft [4, 5]. They are more common in adults, and the proportion of male to female is 8 : 1 [6, 7]. They usually occur within the first year after surgery (time range 1–20 years) [7, 8]. The mortality rate of SAEF has been reported to be up to 50–70% [9].
A recent series [10] demonstrated that SAEF continues to be a life-threatening condition, with a 27% in-hospital mortality rate, which is substantially higher upon emergency presentation, despite prompt diagnosis and treatment. The majority of fistulas involving the intestinal tract are between the aorta and the 3rd–4th portion of the duodenum (between the abdominal aorta and the superior mesenteric artery) in 74% of cases, followed by the small intestine (jejunum more commonly than the ileum) 19%, the colon 5%, and the appendix 1% [11–14].
Pathogenesis
The pathogenesis of SAEFs is not entirely clear although some hypotheses have been described. The main mechanism seems to be the gradual erosion of the aortic graft in the intestine, with or without the presence of inflammation from the intestinal contents. The constant irritation of the intestinal wall by the aortic pulsation seems to play an important role, which explains why most fistulas appear at the proximal anastomotic site of the third portion of the duodenum, which is pressed between the aorta and the superior mesenteric artery in the retroperitoneal space [3, 15–17]. Other hypotheses presuppose the presence of local inflammation and subsequent erosion of the intestinal wall due to infection of the aortic graft during the first operation [11, 15, 18–20]. A positive preoperative blood culture predicts a poor outcome [21, 22].
Clinical picture – diagnosis
The diagnosis of an aortoenteric fistula can become quite challenging, and strong clinical suspicion is required. Although the clinical picture is directly related to the part of the gastrointestinal tract where the fistula has formed, typically patients come with at least one of the following: gastrointestinal bleeding (melena, haematochezia, haematemesis) 80%, sepsis (44%), abdominal pain (30%), lumbar pain 15%, groin mass (12%), and abdominal pulsatile mass (6%). Other underreported symptoms include acute limb ischaemia, pain, vomiting, ileus, and septic emboli to the legs [3, 5, 13]. The typical triad: gastrointestinal bleeding, abdominal pain, and palpable mass, is found in only 6–12% of cases [23]. Intermittent bleeding followed by massive bleeding (Herald bleeding) is typical for the disease and raises a strong suspicion of diagnosis although it occurs in approximately 60% [5, 18, 20, 24, 25]. Song et al. [26] proposed that the occurrence of herald bleeding had a mean frequency of 3.6 episodes. They also found that the time interval between the first herald bleeding and massive exsanguination varied from 5 h to 5 months, with a median of 4 days. More than 50% of cases had a duration longer than 3 days [26]. This time frame can be used as an opportunity for the treatment of fistulas [14].
For imaging, several methods have been proposed, the most useful being oesophagogastroduodenoscopy and computed tomography (CT). The choice of imaging method depends on the patient’s condition, the presence or not of bleeding, and its severity. In the case of massive bleeding, the patient should be taken directly to the operating room without further delay. In acute bleeding in stable patients, endoscopy is usually performed but with low accuracy in the diagnosis of AEF (25–40%) [6, 26, 27]. As already mentioned, most aortoenteric fistulas are found in the 3rd-4th portion of the duodenum and for this reason the endoscope should reach the entire length of the duodenum, while use of an enteroscopy or paediatric endoscopes is recommended [6, 8, 28–30]. The presence of fresh blood and clots are indicative. Colonoscopy is not often cited as a diagnostic method [31, 32].
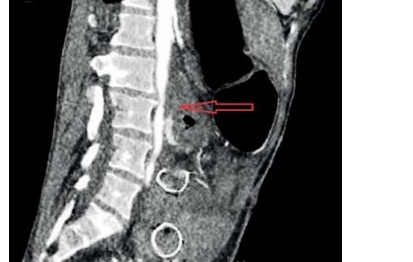
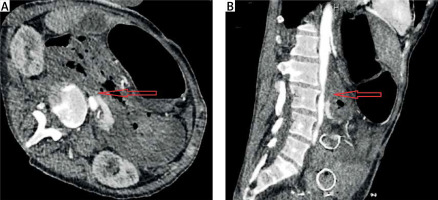
Spiral CT is fast and easily accessible from most centres. Intravenous contrast-enhanced CT helps both in the diagnosis and in the later decision of surgical repair of the aorta due to the anatomical information it provides [25, 33]. CT findings suggestive of AEF are air around the aorta, bowel wall oedema around the aorta, and the loss of a fatty plane between the aorta and GI tract (Figure 2). The fistula itself may not be visualised. CT angiography can be negative, despite an active bleed from an AEF [32]. Despite the great value of imaging, the clinical presence of gastrointestinal bleeding is very important for differentiating AEFs from other entities such as perigraft infection without fistulation.
Treatment
The treatment of SAEFs, regardless of their location, is exclusively surgical, and without it the mortality is 100%. With appropriate treatment, survival rates greatly varying between 18% and 93% have been reported [22, 34]. The choice of technique depends on the patient’s condition, the severity of bleeding, comorbidities, and the presence or absence of inflammation. In the first phase, resuscitation and haemodynamic support are of priority. Preoperative intravenous broad-spectrum antibiotic therapy for Gram+, Gram–, and enteric pathogens may be administrated followed by targeted therapy [35].
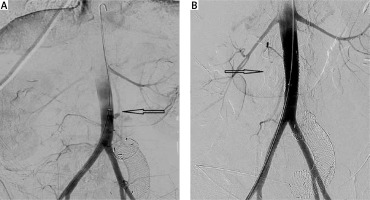
The treatment of AEF includes graft excision and extra-anatomic bypass or in situ graft replacement and simple graft excision alone. Conservative or palliative treatment, utilising antibiotics and drainage or irrigation, is also acceptable for patients unfit for surgery [36]. Endovascular repair is a less invasive alternative, including limited reports for the treatment of AEFs. In SAEF, endovascular treatment (Figure 3) is performed as a bridging therapy to open repair to stabilise the patient until open repair is accessible. A recent multi-centre study [37] for the management of SAEFs found that there was no discernible distinction in all-cause mortality rates among patients who received EVAR or OAR as initial therapy for SAEF. In the acute setting, EVAR may be considered as a primary treatment or a bridge to definitive OAR in conjunction with broad-spectrum antimicrobial therapy for patients with SAEF. Kakkos et al. [13] came to conclusion that for SAEFs, endovascular surgery is associated with a higher early survival rate (p < 0.001) than open surgery, when appropriate, since in hospital mortality was 7.1% for endovascular surgery and 33.9% for open repair. Long-term follow-up erodes the majority of this benefit, because late sepsis occurred more often after endovascular surgery (2-year rate 42% vs. 19% for open, p = 0.001), suggesting that in certain patients, a staged approach with early conversion to in situ vein grafting may produce the best results. Open procedure (staged or not staged) has as its fundamental objectives the confirmation of the diagnosis, the control of bleeding and inflammation, and the restoration of the continuity of the intestinal tract. Older series demonstrate operative mortality as high as 64% [38]. A recent study [39] provides the 20-year experience of a single centre, reporting significant improvement in 30-day (p = 0.03) and 90-day (p = 0.008) mortality even on risk-adjusted analysis, with no significant difference in 1-year survival overall.
Figure 3
A, B – Extravasation of the IV contrast during the aortography and occlusion after the aortic stent placement (black arrows). The migrated duodenal stent is also recognised
The management of aortoduodenal fistulas has been studied more than the other subtypes of aortoenteric fistulas due to the frequency of occurrence. Initially, a laparotomy is conducted with temporary in situ revascularisation with a prosthetic graft, and later, a staged extra-anatomic bypass followed by transabdominal removal of the temporarily placed graft. Duodenal exclusion is not an obligatory adjunct to duodenal repairs, but in some cases resection of the fistula-bearing area of the aneurysm resected en bloc with the attached duodenum may be needed. In some cases extensive destruction of the duodenal wall is noted, and then pylorus-preserving pancreatoduodenectomy is an option. A vascularised pedicle of the greater omentum can be used to wrap around the prosthetic graft or cover the infected surgical field for the purpose of preventing a graft infection. Aortojejunal fistula are rare and usually treated with primary repair, and the jejunal defect is closed using a linear stapler [40, 41]. Aortocolonic fistulas are better treated with some type of colectomy and/ or transverse colostomy. In general, patients who undergo bowel resection have a worse prognosis than those who have simple repair [42]. Iliac-appendiceal fistula is a rare medical entit, which is treated with appendectomy and partial cecectomy [43].
Limitations: As a narrative review, our study has inherent limitations. Our study was done in a systematic way, but it is not a systematic review, and a significant risk of bias remains. Many of the data we have presented may derive from low-quality studies such us case reports. No statistical analysis was done to further interpret the collected data.
In conclusion, the role of the general surgeon in managing an aortoenteric fistula (AEF) is crucial and multifaceted. In short, it can be summarised in the points below:
The surgeon must confirm the diagnosis of SAEF. This is often done through imaging techniques like computed tomography (CT), which provides superior images compared to other diagnostic modalities.
One of the primary responsibilities of the surgeon is to control the bleeding associated with SAEF. This is particularly important because most AEFs are heralded by repetitive gastrointestinal bleeds
The surgeon must repair the damaged area of the bowel
If the condition is caused by an AAA or a previous aortic graft, the surgeon will need to remove the aneurysm or graft that is causing the issue
Once SAEF is diagnosed, preventive measures, such as antibiotic therapy, delicate surgery for eradication of septic focus with thorough debridement of infected and devitalised tissue, and reconstruction of the excised aorta by extra-anatomic or an in situ route, are required
Over time, operative mortality rates have decreased, possibly due to improvements in perioperative care and the advent of endovascular techniques
Aortoenteric fistulas are a rare but fatal gastrointestinal bleeding cause that the general surgeon may be asked to manage. Diagnosis requires the combination of strong clinical suspicion and the presence of a history of AAA surgery. Although a vascular surgery case, general surgeons play a role in choosing the technique of restoring the intestinal tract, which seems to be significantly related to subsequent morbidity and mortality.