Introduction
Hypertrophic cardiomyopathy (HCM) is the most common genetically inherited autosomal dominant cardiac condition, with an estimated prevalence of about 1 in 200 people in the general population. It contributes to substantial morbidity and mortality across all age groups [1]. HCM is a genetically diverse condition with various disease-causing gene mutations and varying phenotypic expressions, leading to different clinical presentations [2]. The first identified HCM mutation was the R403Q mutation in the β-cardiac myosin heavy chain gene (MYH7). Approximately 35% of HCM mutations occur in human β-cardiac myosin, another 35% are found in cardiac myosin-binding protein-C (MyBP-C), which includes 13 domains, and the remaining mutations are distributed across other sarcomeric proteins, primarily in troponins and tropomyosin [3, 4]. Mutations in β-cardiac myosin lead to pathological heart hypertrophy, fibrosis, and disarray of the myofilaments due to increased contractility of the cardiac sarcomere.
Patients with HCM are asymptomatic or experience only mild symptoms, which contributes to the condition often being underdiagnosed [5]. However, for some, HCM can progress to more severe complications, including worsening shortness of breath, chest pain, heart failure (with or without left ventricular systolic dysfunction), atrial fibrillation, and sudden cardiac death (SCD), the latter being the most severe and feared complication [6]. Current treatment guidelines for obstructive HCM primarily emphasize managing symptoms through medications that reduce heart contractility, such as β-blockers, non-dihydropyridine calcium channel blockers, and disopyramide [7]. While these medications help alleviate symptoms for patients with HCM, they do not directly target the underlying pathophysiological mechanisms of the disease, highlighting the need for the development of more advanced therapeutic options. For patients who do not respond adequately to medical treatment and continue to experience significant left ventricular outflow tract (LVOT) obstruction, more invasive interventions such as surgical myomectomy or transcatheter alcohol septal ablation become necessary to relieve the obstruction and improve symptoms, and are recommended for patients with more advanced disease or specific risk factors [7, 8].
The current treatments for HCM primarily provide symptom relief but do not address the underlying disease mechanisms or significantly improve diastolic function. A novel approach involves direct inhibition of cardiac sarcomere contractility, with mavacamten being the first drug of this kind. Mavacamten inhibits cardiac myosin ATPase, reducing actin-myosin cross-bridge formation, which decreases myocardial contractility and enhances ventricular compliance [9]. This targeted mechanism of action represents a significant advancement in managing obstructive HCM. In 2022, based on results from the EXPLORER-HCM trial, the US Food and Drug Administration (FDA) approved mavacamten for patients with symptomatic NYHA class II-III obstructive HCM, marking a major shift in the therapeutic landscape [9].
Building on this, aficamten, a next-generation myosin inhibitor, offers additional benefits, including a shorter half-life for once-daily dosing and faster achievement of steady-state levels. With minimal cytochrome P450 interaction and a wide therapeutic window, aficamten has effectively reduced both resting and provoked LVOT gradients in symptomatic obstructive HCM, as shown in the REDWOOD-HCM trial [10]. This makes aficamten a promising alternative with a favorable safety profile, offering further potential in HCM treatment.
This review aims to assess the efficacy and safety of myosin inhibitors, including mavacamten and aficamten, in treating HCM. By combining data from relevant clinical trials, this study seeks to synthesize and increase the statistical power of available findings, ultimately providing a more precise and comprehensive summary of the clinical impact of these novel therapies in patients with HCM.
Methods
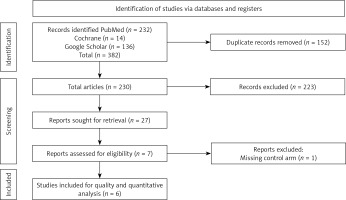
We conducted a thorough literature search using the PubMed, Google Scholar, and Cochrane databases up to September 2024 to identify all randomized controlled trials (RCTs) comparing cardiac myosin inhibitors with placebo in patients with HCM (Supplementary Table SI). Additionally, we examined the references of identified RCTs and prior reviews to uncover further relevant studies. The search adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, ensuring a systematic and rigorous approach to identifying and selecting studies for inclusion in the meta-analysis (Figure 1) [11]. Studies that were case reports, case series, animal studies, systematic reviews, or meta-analyses were excluded from the final analysis. The systematic search yielded articles, which were then imported into EndNote Reference Manager (Version X7.5; Clarivate Analytics, Philadelphia, Pennsylvania). Within EndNote, we systematically screened for and removed any duplicate entries. Two authors (MH and SK) independently screened titles and abstracts, reviewed full-text articles, and determined eligible studies. When eligibility was uncertain, the full text was examined, and any discrepancies were resolved through discussion or consultation with a third reviewer, involving the original authors if necessary. Quality assessment is shown in Supplementary Figure S1.
Eligibility criteria
Inclusion: a) Adult patients (≥ 18 years) diagnosed with either obstructive or non-obstructive HCM, b) RCTs comparing myosin inhibitor to placebo or guideline-based medical treatment for HCM, studies reporting outcomes such as improvement in NYHA class or Kansas City Cardiomyopathy Questionnaire Clinical Summary Score (KCCQ-CSS), d) studies evaluating the safety outcomes, including SAEs and atrial fibrillation onset.
Exclusion: a) Non-randomized studies, including case reports, commentaries, editorials, and animal studies. b) Patients with significant background therapy that could interfere with study results, such as those on β-blockers or diltiazem. c) Studies focusing only on short-term outcomes, incomplete data, or limited follow-up periods without sufficient reporting on long-term safety and efficacy. d) Studies with no control arm.
Data extraction and quality assessment
Two authors (MH and SK) independently reviewed and selected studies and extracted the following data from the eligible articles: baseline characteristics, study type, study year, sample size, age, and gender. These data were entered into a standard Excel sheet and presented in Table I. [9–15]. The primary outcomes of interest were symptomatic improvement, reported as an improvement of at least one NYHA class or more; KCCQ CSS score change from baseline; mean percent change from baseline in LVOT gradient at rest and Valsalva LVOT gradient; percent change from baseline in NT-proBNP, troponin I, and left ventricular ejection fraction (LVEF). The secondary outcomes included any treatment emergent adverse events (TEAEs), such as palpitations, dizziness, nausea, dyspnea, and fatigue during treatment or at follow-up; any serious adverse events (SAEs), including syncope, stress cardiomyopathy, atrial flutter, atrial fibrillation, sinus node dysfunction, systolic dysfunction, arthritis, mental status changes, and renal failure during treatment or at follow-up; and the onset of paroxysmal atrial fibrillation (PAFs) during treatment or at follow-up.
Table I
Summary of included randomized controlled trials evaluating myosin inhibitors in hypertrophic cardiomyopathy
| Author/year | RCT name | Total population | Intervention (n) | Placebo (n) | Age mean (SD) | Gender (M/F) | Different dose | Follow-up duration |
|---|---|---|---|---|---|---|---|---|
| Olivotto et al. 2020 [9] | EXPLORER-HCM | 251 | 123 | 128 | 57.0 (13.5) | 129/122 | 5 mg to titrated dose | 30 weeks |
| Maron et al. [10] | REDWOOD-HCM | 41 | 28 | 13 | 58.2 (12.4) | 25/16 | Titrated by cohort | 10 weeks |
| Coats et al. 2024 [12] | SEQUOIA-HCM | 282 | 141 | 141 | 55.4 (13.1) | 138/144 | 5–20 mg | 24 weeks |
| Tian et al. 2021 [13] | EXPLORER-CN | 81 | 54 | 27 | 51.9 (11.9) | 58/23 | 2.5 mg to titrated dose | 30 weeks |
| Desai et al. 2022 [14] | VALOR-HCM | 112 | 56 | 56 | 60.0 (12.0) | 57/55 | 5–15 mg | 16 weeks |
| Ho Carolyn Y et al. [15] | MAVERICK-HCM | 59 | 40 | 19 | 54.0 (11.2) | 25/34 | 5–10 mg | 16 weeks |
Data analysis
We used the original data to calculate risk ratios (RRs) and their corresponding 95% confidence intervals (CIs) for dichotomous outcomes, applying a random effects model. For continuous outcomes, a meta-analysis of the weighted mean difference (WMD) and its 95% CI was conducted, also using the random effects model to account for variability across studies. To evaluate the level of heterogeneity among the studies, we employed the Higgins I² statistic, categorizing heterogeneity as follows: mild heterogeneity (I²) between 25% and 50%; moderate heterogeneity (I²) between 50% and 75%; severe heterogeneity (I²) exceeding 75% [12]. Subgroup analysis was performed based on the type of myosin inhibitor, aficamten vs. mavacamten. This approach allowed us to effectively account for variations in study designs and populations, ensuring more robust conclusions in our meta-analysis. In all analyses, a p-value of < 0.05 was considered statistically significant. The meta-analyses were performed using STATA statistical software (version 18.5; STATA Corp LP, College Station, TX, USA) to ensure precise and consistent calculations throughout the study. This threshold for statistical significance was applied to determine the robustness of the results across different study outcomes.
Results
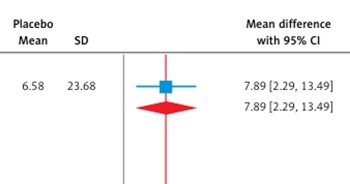
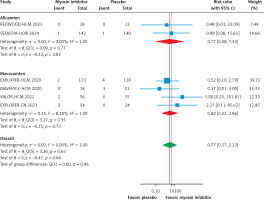
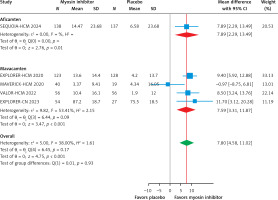
This meta-analysis, which included six RCTs (two evaluating aficamten and four evaluating mavacamten) with a total of 1,081 participants (609 cases and 472 controls), demonstrates that myosin inhibitors significantly improve clinical and biomarker outcomes in patients with HCM compared to placebo. Both agents effectively reduced LVOT gradients, with a pooled mean difference of –70.22 mm Hg (95% CI: –85.42 to –55.03, p < 0.01, Figure 2 A), and showed substantial improvement in the Valsalva LVOT gradient (–61.44 mm Hg; 95% CI: –71.10 to –51.78, p < 0.01, Figure 2 B). Functional status improved significantly, with a pooled RR of 2.21 (95% CI: 1.75 to 2.80, p < 0.01, Figure 3), for achieving at least a one-grade NYHA class improvement. Health status and quality of life, as measured by the KCCQ CSS score, improved significantly, with a pooled mean difference of 7.80 points (95% CI: 4.58 to 11.02, p < 0.01, Figure 4), exceeding the minimal clinically important difference (MCID) of 5 points. Subgroup analysis showed comparable efficacy, with aficamten demonstrating a mean difference of 7.89 points (95% CI: 2.29 to 13.49, p < 0.01, Figure 4) and mavacamten 7.59 points (95% CI: 3.31 to 11.87, p < 0.01, Figure 4).
Figure 2
A – Subgroup analysis forest plot showing the mean difference of LVOT gradient in patients with HCM treated with a myosin inhibitor versus placebo. B – Subgroup analysis forest plot showing the mean difference of Valsalva LVOT gradient in patients with HCM treated with a myosin inhibitor versus placebo

Figure 3
Subgroup analysis forest plot showing the improvement in NYHA class of at least 1 or more in patients with HCM treated with a myosin inhibitor versus placebo
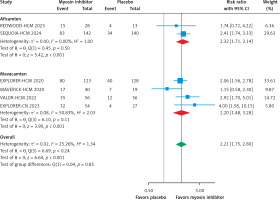
Figure 4
Subgroup analysis forest plot showing the mean difference of KCCQ CSS score in patients with HCM treated with a myosin inhibitor versus placebo
Biomarker analysis revealed significant reductions in NT-proBNP (–69.41 pg/ml; 95% CI: –87.06 to –51.75, p < 0.01, Supplementary Figure S2) and cardiac troponin I (cTnI) (–42.66 ng/l; 95% CI: –48.47 to –36.85, p < 0.01, Supplementary Figure S3), favoring myosin inhibitors. Aficamten demonstrated a greater reduction in LVEF (–10.35%; 95% CI: –13.48 to –7.21, Supplementary Figure S4) compared to mavacamten (–2.50%; 95% CI: –6.21 to 1.20, p > 0.05, Supplementary Figure S4). Safety analysis showed no significant increase in TEAEs (RR = 1.02; 95% CI: 0.92 to 1.14, p = 0.21, Supplementary Figure S5) or SAEs (RR = 0.69; 95% CI: 0.37 to 1.28, p = 0.22, Supplementary Figure S6) compared to placebo. Additionally, atrial fibrillation (Afib) risk was not significantly different between myosin inhibitors and placebo, with a pooled RR of 0.77 (95% CI: 0.27 to 2.23, p = 0.65, Figure 5). Both agents demonstrated consistent efficacy and safety, with subgroup differences in LVEF and cTnI reductions reflecting potential pharmacodynamic variability. These results highlight myosin inhibitors’ efficacy and safety as therapeutic options for symptomatic HCM, improving LVOT gradients, functional capacity, and myocardial biomarkers.
Discussion
We conducted a systematic review and meta-analysis encompassing 6 RCTs with a total of 1,081 participants (609 cases and 472 controls), highlighting the significant efficacy and safety of MIs in managing HCM to evaluate the efficacy of cardiac myosin inhibitors compared to placebo in patients with symptomatic hypertrophic cardiomyopathy. Our findings revealed significant improvements in diagnostic and prognostic biomarkers and transient effects on TEAEs. Reduction in LVOT obstruction was associated with a decreased risk of critical complications, including heart failure, ventricular arrhythmias, cardiac remodeling, and sudden cardiac death. Patients receiving MIs demonstrated notable enhancements in NYHA class and KCCQ-CSS in obstructive and non-obstructive HCM populations. The safety profile of MIs was comparable to that of placebo, with differences in TEAEs but similar rates of SAEs and atrial fibrillation.
Mavacamten, a pioneering allosteric modulator, targets sarcomeric mutations in HCM by selectively inhibiting the cardiac myosin heavy chain, contributing to hypercontractility and impaired myocardial compliance [16]. It reduces dose-dependent contractility by affecting multiple steps in the myosin chemo-mechanical cycle, including phosphatase release and actin-myosin interactions [9, 10, 12–15]. Aficamten similarly binds to cardiac myosin, reducing cross-bridge formation and myocardial contractility, offering a distinct yet comparable therapeutic approach to managing HCM [17, 18].
LVOT obstruction in HCM, defined as a pressure gradient ≥ 30 mm Hg, results from systolic anterior motion (SAM) of the mitral valve, where the anterior leaflet impinges on the hypertrophied septum [19, 20]. This obstruction intensifies during the Valsalva maneuver, exacerbating symptoms such as murmurs and reduced cardiac output [21]. LVOT obstruction is a key predictor of cardiovascular mortality in asymptomatic or mildly symptomatic patients, while NYHA functional class is a stronger marker in those with severe heart failure [22–24]. Prolonged obstruction contributes to cardiac remodeling, increasing risks of myocardial ischemia, arrhythmias, and sudden cardiac death, particularly in young, asymptomatic individuals [25–27]. Supraventricular arrhythmias, particularly atrial fibrillation, occur in approximately 25–30% of HCM patients. These arrhythmias, often secondary to LVOT obstruction and SAM, predispose patients to mitral regurgitation, left atrial enlargement, and subsequent atrial dysfunction [28].
Although cardiac troponins are primarily established as markers of myocyte injury in acute coronary syndromes, several studies have highlighted their prognostic significance in HCM [29]. A 2013 study examining cardiac troponin T levels in HCM patients without coronary artery disease revealed that 54% had elevated troponin levels. During follow-up, 32% of patients with elevated troponin levels, compared to only 7% of those with normal levels, experienced advanced NYHA functional class III/IV heart failure, hospitalizations for heart failure, ventricular arrhythmias, or cardiovascular mortality [30]. Similarly, a 2020 study found that increased serum troponin T concentrations were linked to greater progression of left ventricular remodeling and the development of end-stage dilated cardiomyopathy [31]. Another 2020 study involving 313 HCM patients identified a correlation between elevated troponin I levels. It increased left ventricular wall thickness, severity of LVOT obstruction, and higher fibrosis levels as detected on cardiac magnetic resonance imaging [32].
NT-proBNP, traditionally recognized as a marker of ventricular overload with vasodilatory, diuretic, and natriuretic properties in its active form, has recently been identified as a valuable prognostic indicator in patients with obstructive HCM. Multiple studies have demonstrated a link between elevated levels of pro-BNP and the progression to congestive heart failure and all-cause mortality [33, 34]. A 2022 study conducted by the Cleveland Clinic, involving over 2,100 patients with obstructive HCM, further established an association between NT-proBNP levels and the risk of all-cause mortality or the need for cardiac transplantation [35, 36]. Given their mechanism of action, MIs intentionally reduce LVEF to alleviate LVOT obstruction. However, this effect is dose-dependent and reversible.
In the EXPLORER-HCM trial, five patients experienced protocol-driven temporary treatment discontinuation due to LVEF < 50%, which later normalized and allowed resumption of the trial; 4 other patients had LVEF < 50% at their end-of-treatment visit, with recovery to baseline after 8 weeks in 3 of them. In the MAVERICK-HCM trial, 5 patients in the treatment groups experienced an LVEF decrease to ≤ 45%, leading to drug discontinuation; 4 recovered baseline LVEF within 4 weeks, and the fifth at 12 weeks. In the REDWOOD-HCM trial, 1 patient experienced a transient LVEF reduction to < 50%, which resolved with dose adjustment, with all cohorts returning to baseline within 2 weeks after treatment ended. In the SEQUOIA-HCM trial, 7 patients required dose reductions for LVEF < 50% and 1 had LVEF < 40%, recovering to > 50% at the next visit; all patients returned to baseline after dose adjustments. In the VALOR-HCM trial, four patients had LVEF < 50%, which resolved after drug discontinuation, with recovery occurring promptly within weeks. In the EXPLORER-CN trial, transient LVEF reductions (2.5–5%) occurred, with all cases recovering consistently without long-term adverse outcomes. These findings emphasize the dose-dependent and reversible nature of myosin inhibitors in managing LVEF reductions.
It is important to consider the safety profile of MIs, even though they show promise in treating HCM. Evidence regarding the adverse effects of MIs after or during the intervention remains limited. Our analysis showed no significant differences in SAEs or atrial fibrillation, with a pooled risk ratio of 0.77 (95% CI: 0.27 to 2.23, p = 0.65, Figure 5) for atrial fibrillation, slightly favoring placebo. However, sensitivity analysis for TEAs indicated a pooled risk ratio of 1.02 (95% CI: 0.92 to 1.14, p = 0.21, Supplementary Figure S5), suggesting a minor and inconclusive difference favoring placebo.
The findings are supported by individual trials such as SEQUOIA-HCM and REDWOOD-HCM, which reported no major adverse cardiac events associated with aficamten. Additionally, 8 (5.6%) patients in the aficamten group and 13 (9.3%) patients in the placebo group reported SAEs, none of which were directly attributed to the study drug. In REDWOOD-HCM, one patient in the treatment group had a transient LVEF reduction below 50%, which resolved after dose adjustment, and LVEF returned to baseline within 2 weeks post-treatment.
These findings highlight the dose-dependent and reversible nature of adverse effects related to MIs, particularly reductions in LVEF. However, while no significant long-term safety concerns were identified in trials such as MAVERICK-HCM and VALOR-HCM, their relatively short follow-up durations limit the ability to draw conclusions about long-term safety. Ongoing long-term extension trials, such as those evaluating aficamten and mavacamten, aim to provide more clarity on the extent and duration of adverse effects, thereby enhancing the understanding of their safety profiles in the treatment of HCM.
This meta-analysis has several limitations that should be acknowledged. The included studies had various follow-up durations, with some trials assessing outcomes at 16 weeks and others at 32 weeks, which may have contributed to heterogeneity in the findings. Moreover, the trials primarily compared mavacamten and aficamten to placebo rather than to established medical therapies, restricting the ability to evaluate their relative clinical efficacy. Additionally, two studies focused on aficamten, a cardiac myosin inhibitor with distinct molecular and pharmacokinetic properties compared to mavacamten, which could affect the generalizability of the results. The analysis also did not differentiate between obstructive and non-obstructive hypertrophic cardiomyopathy outcomes due to limited available data. Lastly, although this study provides meaningful insights into the short-term safety and effectiveness of mavacamten, the long-term impacts on important outcomes, such as mortality and the risk of sudden cardiac death, remain insufficiently explored, highlighting the need for future studies with extended follow-up periods.
In conclusion, our meta-analysis highlights the potential clinical and symptomatic benefits of cardiac myosin inhibitors, particularly MIs, in managing symptomatic hypertrophic cardiomyopathy. The treatment demonstrated significant improvements in patient outcomes, such as a reduction of at least one grade in NYHA functional class and enhanced KCCQ CSS scores, without a notable increase in adverse events. Furthermore, our study uniquely assessed the impact of MIs on diverse cardiac imaging parameters, biomarkers such as NT-proBNP and cardiac troponin I, and clinical metrics. This comprehensive evaluation offers a deeper understanding of MI’s effects on the heart’s structural and functional aspects, complementing prior research.