Introduction
Sarcoidosis is a systemic inflammatory disorder characterised by the development of non-caseating granulomas, which are aggregates of immune cells, in various organs throughout the body [1]. Whilst the aetiology is not fully understood, there is a suggestion that environmental stimuli trigger an aberrant immune reaction in genetically susceptible individuals, resulting in granuloma formation [2]. Although sarcoidosis primarily manifests in the pulmonary system, often characterised by symptoms of a persistent dry cough, fatigue, and dyspnoea, its presentation is not limited to the lungs. Other organs, such as the heart, can also be involved [1]. Although myocardial involvement presents symptomatically in approximately 10% of individuals, the associated risk of morbidity and mortality render cardiac sarcoidosis (CS) as a key manifestation of the disease [3, 4]. When there is involvement of the myocardium, the regular electrical conduction may be disrupted and there may be hinderance of the normal cardiac ability to pump effectively. This can result in a range of manifestations including atrioventricular (AV) block, ventricular tachyarrhythmias, and heart failure [4, 5].
ST-elevation myocardial infarction (STEMI) is commonly attributed to atherosclerotic plaque rupture with subsequent occlusion of the coronary artery. Although there are conflicting results between studies, there is research that provides empirical evidence linking a greater risk of STEMI and a diagnosis of sarcoidosis [6–8]. It has been hypothesised that the elevated risk of STEMI in this cohort can be attributed to the alterations in lipid metabolism seen in sarcoidosis, as well as the inflammatory nature of the disease, which both lead to an increased the risk of atherosclerosis [9, 10]. However, to our knowledge there are currently no data available on how the presentation, impact, and outcome of STEMI differs in patients with sarcoidosis versus the general population.
This study aims to address this absence in the literature using propensity-matched real-world data.
Material and methods
Study population
This retrospective study encompassed individuals aged 18 years or older admitted between 2016 and 2020 from the STEMI identified through the International Classification of Diseases, 10th Revision (ICD-10) codes, based on recommendations from previous studies. They were then stratified into groups of individuals with sarcoidosis and without sarcoidosis. The de-identified nature of the NIS waives the need for Ethics and Institutional Review Board approvals. All ICD-10 codes used for our study are available in Supplementary 1 [11–17].
Statistical analysis
Initially, χ2 tests were employed to assess the association between the presence of sarcoidosis in STEMI patients and various categorical variables. These included sex, insurance status, race, smoking status, presence of diabetes, hypertension, cirrhosis, alcohol abuse, drug abuse, peripheral vascular disease (PVD), obesity, chronic kidney disease (CKD), lipid disorder, history of myocardial infarction (MI) or stroke, hospital type, and age group (≥ 60 years vs. < 60 years). This was followed by an evaluation of cardiac arrhythmia events. The main aim of this study is to assess and compare the intersectionality and comorbid status of patients with STEMI who have sarcoidosis and those who do not have sarcoidosis. The second objective of this study is to evaluate a range of outcomes, such as the duration of hospitalisation, acute ischaemic stroke (AIS), coronary artery bypass grafting (CABG), percutaneous coronary intervention (PCI), acute kidney injury (AKI), cardiogenic shock, and mortality, among these specific patient cohorts.
Subsequently, propensity score matching was conducted via R-Studio, adjusting for the aforementioned variables along with age. A 1 : 1 ratio matching with a calliper of 0.2 was utilised to create balanced groups between sarcoidosis and non-sarcoidosis cohorts. A multivariate regression analysis was performed to discern differences in outcomes between the sarcoidosis and non-sarcoidosis groups.
Results
There were 851,290 patients with STEMI recruited in this study, of whom 1215 had sarcoidosis (Table I). Both groups involved mostly males (58.4% vs. 69.5%, p < 0.01) and patients aged ≥ 60 years (64.6% vs. 60.9%, p < 0.01). Patients with sarcoidosis expressed a higher prevalence among females than those without sarcoidosis (41.6% vs. 30.5%, p < 0.01). The prevalence of White and Hispanic individuals within the sarcoidosis cohort was lower (65.3% vs. 75.5% and 3.8% vs. 8.6%, respectively, both p < 0.01), with Black individuals making up a higher proportion (27.1% vs. 8.8%, p < 0.01). Although a marginal difference, sarcoidosis patients were more likely to be admitted to urban teaching hospitals (74.9% vs. 71.9%, p = 0.030).
Table I
Characteristics and outcomes of patients with and without sarcoidosis admitted for ST-elevation myocardial infarction (STEMI) in the United States between 2016–2020
Regarding comorbidities, there was significant variation seen between the 2 groups. Those within the sarcoidosis cohort demonstrated lower rates of smoking (35.4% vs. 52.0%, p < 0.01) and higher occurrences of diabetes (42.4% vs. 32.4%, p < 0.01), cirrhosis (1.6% vs. 0.5%, p < 0.01), depression (9.1% vs. 6.7%, p < 0.01), peripheral vascular disease (7.8% vs. 5.1%, p < 0.01), obesity (24.3% vs. 17.8%, p < 0.01), chronic kidney disease (25.9% vs. 13.1%, p < 0.01), lipid disorder (71.2% vs. 64.6%, p < 0.01), and prior stroke (7.4% vs. 5.2%, p < 0.01). However, there were no statistically significant differences observed between the groups for hypertension (45.3% vs. 47.7%, p = 0.090), alcohol abuse (2.1% vs. 3.5%, p = 0.091), old myocardial infarction (11.9% vs. 11.9%, p = 0.976), or mean Charlson Comorbidity Index (CCI) score (3.14 vs. 2.68, p = 0.652). There were no significant differences in the occurrence of several cardiac arrhythmias, such as atrial fibrillation (15.6% vs. 14.0%, p = 0.106), supraventricular tachycardia (SVT) (2.5% vs. 2.2%, p = 0.463), ventricular tachycardia (11.9% vs. 12.6%, p = 0.456), ventricular fibrillation (11.1% vs. 11.1%, p = 0.969), or atrial flutter (1.2% vs. 1.7%, p = 0.231).
Rates of PCI in the sarcoidosis cohort were lower than those for non-sarcoidosis patients (75.3% vs. 79.8%, p < 0.01), and the patients were more likely to have an increased mean length of stay (LOS) (4.76 vs. 4.01 days, p < 0.01). Sarcoidosis patients had a significantly higher incidence of acute kidney injury (AKI) (23.5% vs. 17.1%; p < 0.01); however, there were no significant differences between the sarcoidosis and non-sarcoidosis groups in rates of coronary artery bypass graft (CABG), acute ischaemic stroke (AIS), cardiogenic shock, or mortality.
After propensity score matching (PSM), 1180 patients were assessed in each cohort, maintaining balance in characteristics and co-morbid conditions (Table II). Accordingly, comparable distributions were observed in demographic attributes, comorbidities, and hospital characteristics between sarcoidosis and non-sarcoidosis patients. The results demonstrate that the only notable difference relating to outcome was having lower rates of CABG seen in the sarcoidosis cohort (5.5% vs. 8.5%, p = 0.014). Previously observed differences in the incidence of AKI, PCI, and mean LOS were no longer evident after PSM. Concerning the aforementioned cardiac arrhythmias, SVT was observed significantly more in the sarcoidosis group (2.5% vs. 1.3%, p = 0.024), whilst the other arrhythmias were seen in similar distributions between the 2 cohorts.
Table II
Characteristics and outcomes of patients with and without sarcoidosis admitted for STEMI in the United States between 2016 and 2020 after propensity score matching
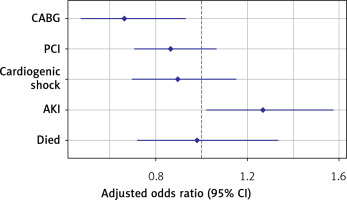
In the adjusted outcomes analysis (Table III, Figure 1), sarcoidosis was associated with a lower incidence of CABG (adjusted odds ratio [aOR] = 0.663; 95% confidence interval [CI]: 0.472–0.931, p = 0.018), and high rates of AKI (aOR = 1.269, 95% CI: 1.02–1.58, p = 0.033). Conversely, the presence of sarcoidosis had no significant effect on the incidence of PCI, cardiogenic shock, mortality, and LOS.
Table III
Adjusted odds ratio of events in patients with and without sarcoidosis (reference) admitted for STEMI in the United States
| Parameter | P-value | aOR | Lower 95% CI | Upper 95% CI |
|---|---|---|---|---|
| CABG | 0.018 | 0.663 | 0.472 | 0.931 |
| PCI | 0.174 | 0.866 | 0.705 | 1.065 |
| Cardiogenic shock | 0.392 | 0.896 | 0.696 | 1.153 |
| AKI | 0.033 | 1.269 | 1.020 | 1.578 |
| Died | 0.899 | 0.980 | 0.720 | 1.335 |
Discussion
The primary findings from our analyses of this large, nationwide data set were threefold. Firstly, other than the lower rates of smoking seen in the sarcoidosis cohort, STEMI patients with co-existing sarcoidosis generally presented with a greater burden of co-morbidities when compared to the non-sarcoidosis group. Secondly, sarcoidosis was associated with an increased incidence of SVT and AKI. Finally, those within the sarcoidosis group had a lower incidence of CABG post-STEMI.
Patient characteristics
This study demonstrated significant disparities in patient characteristics between the 2 groups. Overall, there were more male patients with STEMI in both of our cohorts, which can be attributed to the well-documented difference in the prevalence of myocardial infarction between sexes [18]. However, when looking at each subgroup, there was a higher proportion of females seen in the sarcoidosis group compared to the non-sarcoidosis group. This finding is in keeping with previous research which shows a greater incidence of sarcoidosis in females [19].
Our research findings also showed significant racial disparities between the 2 groups, in particular relating to the greater proportion of Black individuals seen within the sarcoidosis cohort. Again, this finding is consistent with existing data, such as the review of 9 studies from Hena which showed a tendency for sarcoidosis to affect Black individuals [20]. More specifically, this review showed that Black females were at greatest risk of morbidity and mortality associated with sarcoidosis – a finding also replicated from studies not just in the United States, but across the world [19].
The analysis also investigated insurance patterns amongst those with sarcoidosis. Medicaid is a joint federal and state initiative that provides healthcare insurance coverage to households with low incomes [21]. In our study, those with Medicaid showed lower prevalence of STEMI, with the converse being true of those with private insurance. This may indicate a correlation between STEMI occurrence in sarcoidosis patients and a higher socio-economic status. Although there is limited evidence to support or refute this finding, reports suggest that in those with sarcoidosis it is in fact lower socioeconomic status that is associated with worsened morbidity and mortality [22].
Co-morbidities
We demonstrated that individuals diagnosed with sarcoidosis were less likely to be smokers but had higher prevalence rates of diabetes, cirrhosis, depression, peripheral vascular disease, obesity, and chronic kidney disease.
Several previous studies suggest that smoking may be a protective factor against developing pulmonary sarcoidosis but may elevate the risk of extrapulmonary manifestations [23, 24]. However, as is true of many presentations of sarcoidosis, geoepidemiological variations between populations are associated with diverse and varied manifestations. For instance, although a Japanese study showed higher rates of pulmonary sarcoidosis with a lower incidence of cutaneous involvement in current smokers [25], a study from India showed no significant link between smoking and sarcoidosis [26]. Blanchet et al. investigated the impact of nicotine therapy on hypersensitivity pneumonitis, a different disease also characterised by granulomatous formation. Although this study investigated hypersensitivity pneumonitis, the outcomes from this research potentially have broader implications for other granulomatous diseases such as sarcoidosis. This study showed nicotine therapy to reduce white blood cell counts across all cell types, sparing CD8 lymphocytes, but more relevantly to our study – CD4 lymphocytes as well [27]. Although CD4+ lymphocytes play a central role in the development of granuloma formation, and consequently sarcoidosis, if these are unaffected by nicotine as extrapolated from this study, then this would imply that other white blood cells may be involved in the pathogenesis of the disease. This would result in a reduced incidence of sarcoidosis in those who smoke, a finding in keeping with the results seen in our study [2]. However, this postulation and result is hypothesis generating at best.
Our study showed those with sarcoidosis had high incidences of metabolic syndromes such as diabetes and obesity. Previous studies, such as a systematic review from 2021, also showed statistical significance linking sarcoidosis and diabetes [28]. Other prior studies from Cozier et al. (2014) and Ungprasert et al. (2016) correlated sarcoidosis with obesity, with the authors postulating that this relationship could be explained by the presence of increased levels of leptin, an immunomodulatory hormone that is more readily secreted in obese individuals [29, 30]. In turn, leptin induces immune cell proliferation, in particular upon the naive T-cells, which triggers a shift towards Th1 immunity – a process that may be implicated in the development of sarcoidosis [31].
The elevated depression rates seen in our study also align with previous investigations by Hinz et al. (2012), Goracci et al. (2008), and Chang et al. (2001) [32–34]. However, due to the absence of sufficient data pertaining to the temporal relationship between the development of this condition and the manifestation of sarcoidosis, there is ambiguity regarding the causality of this association.
One such explanation for this relationship may lie in the treatment of sarcoidosis using immunosuppression. Side effect profiles of these medications leading to symptoms such as neuropsychological disturbances may account for the higher rates of depression seen in this cohort, with other Cushing’s syndrome features such as central adiposity and insulin resistance leading to obesity and diabetes [1, 35]. Further research is warranted to elicit whether these associations between sarcoidosis, metabolic syndromes, and depression are in fact causal or are of a consequence of glucocorticoid therapy.
The current study also demonstrated higher prevalence of liver cirrhosis among individuals with sarcoidosis, which is consistent with findings from small-scale studies and case reports [36–41]. Cirrhosis in sarcoidosis patients has been hypothesised to be a direct consequence of granuloma formation in the liver [41]. Similarly PVD, another comorbidity variable which reached significance in the sarcoidosis cohort, has been plausibly linked to vasculitic lesions in the peripheral vessels due to granulomatous inflammation [42]. Although existing literature indicates a propensity for sarcoidosis to impact renal function in the context of acute renal injury, our study also found that CKD emerged as a comorbidity seen more frequently in the sarcoidosis cohort [43–45]. The association between sarcoidosis and chronic renal disease, liver cirrhosis, and PVD are scarcely discussed in the literature, and although we have established a significant correlation, further research is necessary to establish an accurate narrative.
Cardiac events and arrhythmias
We expected that, as a consequence of the highly arrhythmogenic nature of cardiac sarcoidosis (CS), there would be a link between sarcoidosis patients in this cohort and arrhythmia. After propensity matching, our analysis showed significance linking supraventricular tachycardia (SVT) and sarcoidosis, in keeping with our hypothesis. Desai et al. (2016) observed arrhythmias, primarily atrial fibrillation, in around 20% of sarcoidosis patients [46], with a subsequent study from Yasuda et al. (2016) identifying ventricular tachyarrhythmias as the predominant cardiac event observed in individuals diagnosed with cardiac sarcoidosis [47]. A more recent meta-analysis from Mahmoud et al. (2020) highlighted unspecified arrhythmias as the third most prevalent cardiac comorbidity in sarcoidosis cases [48].
Notably, without propensity matching, we found no correlation between cardiac arrhythmias and sarcoidosis. We suspect this may be due to confounding factors, primarily age. A study conducted by Piccini et al. showed escalating incidences of atrial fibrillation with respect to increasing age, specifically among individuals aged 65 years and above [49]. On a similar note, the previously discussed study by Desai et al. (2016) observed a statistically significant difference in age between the sarcoidosis cohort with arrhythmias (mean age 61.9 years) and the sarcoidosis cohort without arrhythmias (mean age 56.0 years) [46]. As such, it can be stated that the incidence of arrhythmias increases with age regardless of the presence of sarcoidosis. The mean ages for both groups in our study are relatively similar, and therefore the absence of significant variation between groups may not reflect reduced arrhythmia rates in the sarcoidosis group, but rather increased rates of arrhythmia in the non-sarcoidosis group due to the elderly nature of the population observed in our study.
Outcomes
PCI is widely regarded as the preferred therapeutic approach for most cases involving STEMI [50]. CABG is a major surgical operation reserved for cases not amenable to PCI [51]. While our study indicates decreased rates of CABG and PCI in sarcoidosis, there are few data specifically addressing STEMI treatment in sarcoidosis, so these findings could not be corroborated [52]. Further research observing the type of coronary artery vessels, the number of arteries, and the type of lesions involved are needed to draw meaningful conclusions.
Notably, sarcoidosis patients had higher AKI rates, aligning with prior studies outlining the occurrence of AKI in sarcoidosis [45, 53–55]. STEMI leads to a reduction in cardiac output, resulting in a pre-renal injury to the kidneys [56]. In the sarcoidosis group, AKI may have occurred more frequently due to the compounding effects of STEMI superimposed onto the granulomatous interstitial nephritis and hypercalcaemia thought to cause AKI in sarcoidosis patients [55, 57, 58].
Strengths and limitations
This study’s primary strength lies in its extensive sample size, offering diverse representations reflective of the target population. Furthermore, the study is robust due to minimising selection bias. However, its retrospective nature limits the comprehensiveness of the data. It prevents us from truly establishing clear temporal relationships between variables and challenging accurate causality inferences. Moreover, adjustments were limited to collected variables, potentially missing other confounding factors specific to this study. Lastly, the data’s exclusive focus on the United States warrants caution in generalising findings to other populations, because well-documented demographic variations between those with sarcoidosis may not be captured by our results.
In conclusion, STEMI patients with sarcoidosis carry a higher burden of comorbidities compared to those without sarcoidosis. Additionally, although we identified STEMI patients with co-existing sarcoidosis to exhibit worsened outcomes, it is important to note that these outcomes are probably influenced by confounding variables associated with sarcoidosis rather than being a direct consequence of the condition itself. Using a large, propensity matched, real-world dataset of STEMI patients, we showed sarcoidosis to be associated with fewer cases of CABG and a greater incidence of AKI and SVT when compared to non-sarcoidosis patients.