Introduction
Patients with atherosclerosis receive oral antiplatelet therapy for a long time. Aspirin is the most commonly used antiplatelet agent to prevent thrombo-embolic events [1, 2]. However, some patients have a reduced or absent response to aspirin, which is referred to as aspirin resistance (AR) [3, 4]. With laboratory methods AR could be defined as the inability of aspirin to inhibit in vitro platelet activity [5, 6]. Clinically, AR has been defined as the failure of aspirin to prevent thrombo-embolic vascular events despite regular therapy [6, 7]. It has been reported that AR increases the risk of recurrent ischemic vascular events [7, 8]. The predictors of AR have not been determined yet. Clinical, pharmacological and genetic factors may be associated with AR [8]. Mechanisms of resistance to this commonly used drug should continue to be investigated, as it can have significant consequences when ineffective. Previous studies have reported that the frequency of AR varies widely, from 5% to 45% [9].
Vitamin D (VD) deficiency is an important health problem that concerns a large population. VD has been found to have a wide variety of biological effects. Moreover, it has been shown to be associated with cardiovascular disorders and thrombotic risk [10, 11]. Besides the regulation of calcium and phosphorus levels, VD receptors have also been identified on the surface of cardiomyocytes, smooth muscle cells, inflammatory cells and even platelets. These data indicate that VD may regulate platelet aggregation [12–14]. Based on this hypothesis, we aimed to investigate the relationship between VD deficiency and AR in patients with stable coronary artery disease (CAD) using aspirin and to identify predictors of AR.
Material and methods
We included 70 consecutive patients admitted to the cardiology outpatient clinic, who had been using aspirin (100 mg/day) for at least 7 days due to stable CAD. Blood samples were obtained from all patients to evaluate AR and VD deficiency.
All other clinical conditions that may effect platelet function were determined as the exclusion criteria. In this respect, those who had used anticoagulants or other antiplatelet drugs or nonsteroidal anti-inflammatory drugs in the last 10 days, those with severe renal or hepatic failure, malignancy, alcohol abusers, those with hematocrit < 35% or > 50% or platelet count < 150,000, with bleeding disorders, and those who had had a major surgical procedure within 1 month and a new cardiovascular event within 3 months were excluded from the study.
The study was approved by the local ethical committee, and all patients gave informed consent.
Biochemical measurements
For the measurement of 25-hydroxyvitamin D [25-(OH)D], 3 ml of fasting blood was taken from all patients and placed in two test tubes containing EDTA. Tubes were stored at –70°C within 30 min of collection. Serum 25-(OH)D concentration was measured using an automated Vitamin D2-D3 High Performance Liquid Chromatography (HPLC) Analyzer (Zivak Technologies 25-OH-Vitamin D2-D3 HPLC analysis kit). All blood samples were collected during winter (November–February); therefore the patients with 25-(OH)D level < 20 ng/ml were considered as VD deficient according to Endocrine Society guidelines [15].
Platelet function assessment
Fasting blood samples (3 ml) were collected from each patient into test tubes containing hirudin. Within 1–2 h after blood samples were collected, the platelet aggregation response was analyzed with the Multiplate Platelet Function Analyzer (Multiplate) system. This device is based on the classical whole blood impedance aggregometer. The device has 5 channels for parallel detections and an internal computer system for real-time analysis and documentation. The analysis is based on platelet adhesion upon activation, which causes platelets to aggregate on metal sensor wires in the test cell. For the test, 3 ml of blood anticoagulated with hirudin and the agonist arachidonic acid are added. The ability of platelets to adhere to metal sensors is determined for the next 6 min. The adhesion and aggregation of platelets were measured by the electrical resistance change between two sensor wires. The increase of impedance by the attachment of platelets onto the Multiplate sensors is transformed to arbitrary aggregation units (AU) and plotted against time. The most important parameter is the area under the aggregation curve (AUC), which is best suited to express overall platelet activity. The unit of the AUC is AU*min (as the y-axis is the aggregation, expressed in aggregation units (AU), and the x-axis is the time, expressed in minutes). The AUC can be expressed in U (1 U corresponds to 10 AU*min). Patients were defined as aspirin-sensitive (AS) when their AUC was ≤ 30 U, and with AR when their AUC was > 30 U [16–19].
Statistical analysis
Statistical analysis was performed with SPSS 21.0 for Windows (SPSS Statistics IBM®). Descriptive statistics were obtained for all study variables. Continuous variables were presented as mean ± standard deviation, and categorical variables were presented as frequencies and percentages. The Pearson correlation coefficient and Spearman rank correlation coefficient were used for linear correlation analysis. The significant differences between groups were evaluated using the χ2 test (when variables were categorical) or the independent-samples t test (when variables were continuous). The Mann-Whitney U test was used to compare differences in median values between the patient groups. For the VD level, the receiver operating characteristic (ROC) curve was obtained, and the optimal value with the greatest total sensitivity and specificity in the prediction of AR was selected. A p-value < 0.05 was considered statistically significant.
Results
Seventy patients with stable CAD who had been using 100 mg of aspirin for at least 7 days were included in the present study. The mean age of the study population was 60.2 ±8.3 years. The study population was male-dominant (64%, n = 45). The average duration of aspirin use was 59.3 ±41.3 months. AUC was > 30 U in 15 (21%) patients and these patients were considered AR. The mean AUC was 66.8 ±24.7 U in the AR group and 13.4 ±7.1 U in the AS group.
The mean 25-(OH)D level was 18.7 ±12.2 ng/ml in all patients. Forty-five (64%) patients with 25-(OH)D level < 20 ng/ml were VD deficient and 25 (36%) patients with 25-(OH)D level ≥ 20 ng/ml were VD sufficient. The rate of AR was higher in the VD deficient group than the sufficient group (29%, n = 13 vs. 8%, n = 2, p = 0.041). The mean AUC value was higher in the VD deficient group than the sufficient group (30.2 ±29.1 U vs. 15.3 ±13.1 U; p = 0.018). There were no significant differences in the distribution of age, gender, body mass index, smoking, hypertension (HTN), diabetes or biochemical indexes between the VD deficient and sufficient groups. The demographic, clinical, and laboratory features of the VD deficient and sufficient groups are listed in Table I.
Table I
Demographic, clinical, and laboratory features of VD sufficient and deficient patients
[i] BMI – body mass index, HTN – hypertension, DM – diabetes mellitus, Hgb – hemoglobin, WBC – white blood cells, HDL – high-density lipoprotein, LDL – low-density lipoprotein, ACE – angiotensin converting enzyme, ARB – angiotensin receptor blocker, CCB – calcium channel blocker, PPI – proton pump inhibitor, AUC – area under curve, 25-(OH)D – 25-hydroxyvitamin D.
The relationship between AUC and clinical, demographic features, biochemical indexes and 25-(OH)D level were evaluated via Spearman’s or Pearson’s correlation analyses. The negative correlation of VD level with AUC did not reach statistical significance (r = –0.225, p = 0.062). HT was negatively correlated with AUC (r = –0.303, p = 0.011). There were no correlations between AUC and age, sex, smoking, DM and lipid parameters (Table II).
Table II
Correlation of area under curve (AUC) and 25-(OH)D level with clinical and laboratory parameters
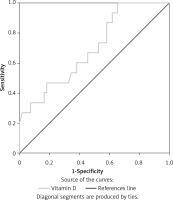
In ROC analysis 25-(OH)D level < 19.25 ng/dl predicted AR with 86.7% sensitivity and 61.8% specificity (AUC = 0.696, 95% CI: 0.551–0.840, p = 0.021) (Figure 1).
Figure 1
ROC curve analysis showing the specificity and sensitivity of the 25-(OH)D level in predicting aspirin resistance
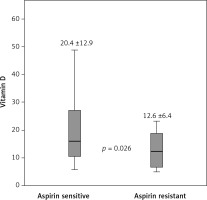
The mean 25-(OH)D level was significantly lower in the AR group than the AS group (12.6 ±6.4 vs. 20.4 ±12.9 ng/dl, p = 0.026) (Figure 2). The frequency of HT was significantly higher in the AS group than the AR group (91%, n = 50 vs. 60%, n = 9, p = 0.003). Female gender ratio was higher and DM was more common in the AR group than the AS group but these differences were not statistically significant (53% vs. 33%; 60% vs. 49%, respectively). There was no difference between the AR and AS groups in terms of lipid parameters or smoking.
Discussion
Seventy consecutive patients with stable CAD who had been using 100 mg aspirin for at least 7 days were included in the present study. The platelet aggregation response was analyzed using the Multiplate system. The most important results of our study can be summarized as follows: 15 (21%) patients had AR and 45 (64%) had VD deficiency. The AR rate and mean AUC value were higher in the VD deficient group than the sufficient group. HT was significantly negatively correlated with AUC, while the negative correlation of VD level with AUC was not significant. The mean 25-(OH)D level was lower in the AR group than the AS group. As far as we know, there is no other study directly examining the relationship between VD deficiency and AR detected by the Multiplate method.
As an antiplatelet drug, the long-term intake of 75–150 mg/day of aspirin reduces the incidence of nonfatal myocardial infarction, cerebral infarction, and fatal cardiovascular events by 32% [20, 21]. However, some patients on chronic aspirin treatment still experience ischemic events [22]. In a prospective study, 326 patients with stable CAD, on aspirin treatment (325 mg/day for at least 7 days) were enrolled for 2 years. Aspirin sensitivity was measured by the optical platelet aggregation method. During the follow-up, AR was associated with a threefold increase in the risk of major adverse events (death, myocardial infarction and cerebrovascular accident) compared with patients who were AS (24% vs. 10%, p = 0.030) [23].
The prevalence and clinical predictors of AR were examined in a prospective study in 325 patients with stable CAD who were receiving aspirin (325 mg/day for at least 7 days). Platelet functions were evaluated and the rate of patients with AR was 5.5% with optical aggregation and 9.5% with PFA-100 methods. It was found that AR increased with age and was more common in women [24]. The higher rate of AR in our study may be due to the difference in the method used. In addition, the use of high-dose aspirin in that study may have contributed to the decrease in AR frequency. In our study, no relationship was found between age and AR. The rate of female gender was higher in patients with AR, but this was not statistically significant. Different studies have reported rates of AR detected by different methods in different clinical situations. The prevalence of AR has been detected as 29.6% by the PFA-100 in 98 patients with stable CAD on 160 mg daily aspirin treatment [25].
Chen et al. aimed to determine whether there was an association between AR and concomitant cardiovascular medication. Using the PFA-100 method, AR was evaluated in aspirin-treated (100 mg/day for at least 2 weeks) patients. AR was detected in 147 (17.7%) of 831 patients. They demonstrated that concomitant ARB treatment in aspirin-treated patients decreased the risk of AR [26]. Supporting the findings of this study, presence of HTN was negatively correlated with AUC in our study. In other words, the frequency of HTN was higher in patients who were AS, and the percentage of ARB use was higher in relation to this. Some prior studies documented that there was a higher prevalence of AR in patients with DM than those without [27]. In our study, the frequency of DM was higher in patients with AR but this was not statistically significant.
VD is a fat-soluble vitamin and an important component of bone and mineral metabolism [28]. VD is also a hormonal factor that has been determined to have an effect on cardiovascular function [29, 30]. It has been shown to prevent endothelial dysfunction and inflammatory processes [31], which form the pathophysiological basis of atherosclerosis [32]. VD deficiency has been associated with increased mortality and cardiovascular risk [10, 11]. Interest in the role of VD in thrombotic risk has increased since the identification of the VD receptor (VDR) on platelets [12, 13]. This suggests that the function of platelets can be affected by the VD level. By now, a few studies about the association between VD level and platelet activation have been reported. In a previous study, it was found that platelet aggregation induced by ADP was decreased in patients treated with calcitriol [33]. It has also been reported that VD level was negatively correlated with mean platelet volume and associated with increased incidence or risk of acute coronary syndrome, HTN and stroke in patients with stable CAD [34]. Increased platelet aggregation and thrombosis risk have been demonstrated in VDR null mice [35]. In a study investigating the AR mechanism in 38 patients with stable CAD, three different isotypes of VD binding protein were found to be higher in patients with AR [36]. This study reveals a genetic predisposition associated with VD in the AR mechanism. Based on these data, it can be hypothesized that VD concentration is associated with platelet aggregation and AR.
In our study, we found that the frequency of AR was increased in patients with VD deficiency. The frequency of AR appears to be quite high, and when not detected, it can have serious clinical consequences. It is very important to detect AR, to determine the mechanisms that cause AR, and to treat it if possible. VD supplementation can reduce platelet aggregation and overcome resistance to aspirin. The true mechanisms remain unclear, and further study is needed. The study had limitations. Firstly the study group was relatively small. In addition, we did not collect data at long-term follow-up; therefore we could not evaluate whether AR was related to the occurrence of cardiovascular events in our population.
In conclusion, in the present study, an association was found between VD deficiency and AR in patients with stable CAD. Detection and treatment of VD deficiency may be useful in preventing AR. More comprehensive studies are needed to determine the effect of VD level on platelet functions and AR.